Development of spray dried liposomal dry powder inhaler of Dapsone
- PMID: 18446460
- PMCID: PMC2976880
- DOI: 10.1208/s12249-007-9024-6
Development of spray dried liposomal dry powder inhaler of Dapsone
Abstract
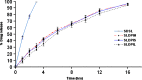
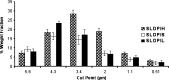
This investigation was undertaken to evaluate practical feasibility of site specific pulmonary delivery of liposomal encapsulated Dapsone (DS) dry powder inhaler for prolonged drug retention in lungs as an effective alternative in prevention of Pneumocystis carinii pneumonia (PCP) associated with immunocompromised patients. DS encapsulated liposomes were prepared by thin film evaporation technique and resultant liposomal dispersion was passed through high pressure homogenizer. DS nano-liposomes (NLs) were separated by ultra centrifugation and characterized. NLs were dispersed in phosphate buffer saline (PBS) pH 7.4 containing different carriers like lactose, sucrose, and hydrolyzed gelatin, and 15% L-leucine as antiadherent. The resultant dispersion was spray dried and spray dried formulation were characterized to ascertain its performance. In vitro pulmonary deposition was assessed using Andersen Cascade Impactor as per USP. NLs were found to have average size of 137 +/- 15 nm, 95.17 +/- 3.43% drug entrapment, and zeta potential of 0.8314 +/- 0.0827 mV. Hydrolyzed gelatin based formulation was found to have low density, good flowability, particle size of 7.9 +/- 1.1 microm, maximum fine particle fraction (FPF) of 75.6 +/- 1.6%, mean mass aerodynamic diameter (MMAD) 2.2 +/- 0.1 microm, and geometric standard deviation (GSD) 2.3 +/- 0.1. Developed formulations were found to have in vitro prolonged drug release up to 16 h, and obeys Higuchi's Controlled Release model. The investigation provides a practical approach for direct delivery of DS encapsulated in NLs for site specific controlled and prolonged release behavior at the site of action and hence, may play a promising role in prevention of PCP.
Figures


Similar articles
-
Nano-liposomal dry powder inhaler of tacrolimus: preparation, characterization, and pulmonary pharmacokinetics.Int J Nanomedicine. 2007;2(4):675-88. Int J Nanomedicine. 2007. PMID: 18203434 Free PMC article.
-
Nano-liposomal dry powder inhaler of Amiloride Hydrochloride.J Nanosci Nanotechnol. 2006 Sep-Oct;6(9-10):3001-9. doi: 10.1166/jnn.2006.405. J Nanosci Nanotechnol. 2006. PMID: 17048511
-
Development of Novel Chitosan Microcapsules for Pulmonary Delivery of Dapsone: Characterization, Aerosol Performance, and In Vivo Toxicity Evaluation.AAPS PharmSciTech. 2015 Oct;16(5):1033-40. doi: 10.1208/s12249-015-0283-3. Epub 2015 Feb 5. AAPS PharmSciTech. 2015. PMID: 25652730 Free PMC article.
-
Lung delivery of nanoliposomal salbutamol sulfate dry powder inhalation for facilitated asthma therapy.J Liposome Res. 2019 Dec;29(4):332-342. doi: 10.1080/08982104.2018.1531022. Epub 2019 May 23. J Liposome Res. 2019. PMID: 30296863
-
Redispersible liposomal-N-acetylcysteine powder for pulmonary administration: development, in vitro characterization and antioxidant activity.Eur J Pharm Sci. 2014 Dec 18;65:174-82. doi: 10.1016/j.ejps.2014.09.017. Epub 2014 Sep 28. Eur J Pharm Sci. 2014. PMID: 25263567
Cited by
-
Dry Powder for Pulmonary Delivery: A Comprehensive Review.Pharmaceutics. 2020 Dec 28;13(1):31. doi: 10.3390/pharmaceutics13010031. Pharmaceutics. 2020. PMID: 33379136 Free PMC article. Review.
-
Aerosol Delivery of siRNA to the Lungs. Part 2: Nanocarrier-based Delivery Systems.Kona. 2017;34:44-69. doi: 10.14356/kona.2017005. Epub 2016 Apr 30. Kona. 2017. PMID: 28392618 Free PMC article.
-
Carrier Effect in Development of Rifampin Loaded Proliposome for Pulmonary Delivery: A Quality by Design Study.Adv Pharm Bull. 2022 Mar;12(2):336-345. doi: 10.34172/apb.2022.032. Epub 2021 May 18. Adv Pharm Bull. 2022. PMID: 35620341 Free PMC article.
-
Aerosol Delivery of siRNA to the Lungs. Part 1: Rationale for Gene Delivery Systems.Kona. 2016 Feb 28;33:63-85. doi: 10.14356/kona.2016014. Epub 2015 Sep 30. Kona. 2016. PMID: 27081214 Free PMC article.
-
Nano-in-Microparticles for Aerosol Delivery of Antibiotic-Loaded, Fucose-Derivatized, and Macrophage-Targeted Liposomes to Combat Mycobacterial Infections: In Vitro Deposition, Pulmonary Barrier Interactions, and Targeted Delivery.Adv Healthc Mater. 2022 Jun;11(11):e2102117. doi: 10.1002/adhm.202102117. Epub 2022 Feb 18. Adv Healthc Mater. 2022. PMID: 35112802 Free PMC article.
References
-
- Murray J. F., Mills J. Pulmonary infectious complications of human immunodeficiency virus infection. Part I. Am. Rev. Respir. Dis. 1990;141(5 Pt 1):1356–1372. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

