Formulation and evaluation of microemulsion based delivery system for amphotericin B
- PMID: 18446472
- PMCID: PMC2976899
- DOI: 10.1208/s12249-007-9022-8
Formulation and evaluation of microemulsion based delivery system for amphotericin B
Abstract
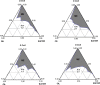
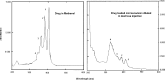
The present studies were designed to develop a formulation of amphotericin B in a lipid-based preparation as a microemulsion and to compare its toxicity with the commercial formulation Fungizone. The final product developed is a lyophilized amphotericin B, oil and surfactant blend for reconstitution in water to yield a microemulsion containing 5 mg/ml of the drug. Pseudoternary phase diagrams were constructed to identify areas of existence of microemulsion composed of Peceol (glyceryl monooleate) as oil phase and Mys 40 (polyethylene glycol 40 stearate) and Solutol HS 15 (polyethylene glycol 15 hydroxy stearate) as surfactants. Amphotericin B was co-evaporated with oil - surfactant mixture to produce a microemulsion pre-concentrate. The co-evaporate was diluted in water, filtered for sterilization and lyophilized to obtain the final product. The lyophilized as well as the reconstituted products were separately studied for stability and the latter was also characterized for various physicochemical aspects including droplet size of the dispersed phase, osmolarity and aggregation state of drug. The dispersion showed no evidence of precipitation of drug for 48 h, and resisted destabilization due to freeze-thaw cycles or centrifugation. The dispersed phase globules measured a mean size of 84 nm and uv-spectrophotometric studies indicated the presence of self-aggregated amphotericin B. The present formulation showed a 92% decrease in haemolysis of human RBC in vitro when compared with the commercially available Fungizone. The LD(50) in mice was estimated to be 3.4 mg/kg. The results indicate that the formulation holds promise for development as a safer and efficacious alternative for amphotericin B therapy.
Figures

References
-
- Adedayo A., Bernardo J. F., Swenson C. E., Bolsack L. E., Horwith G., DeWit S., Kelly E., Klasterksy J., Sculier J. P., DeValeriola D., Anaissie E., Lopez-Berestein G., Llanos-Cuentas A., Boyle A., Branch R. A. Pharmacokinetic profile of ABELCET (amphotericin B lipid complex injection): combined experience from phase I and phase II studies. Antimicrob. Agents Chemother. 1997;41(10):2201–2208. - PMC - PubMed
-
- Anya M. H. Supramolecular lipid drug delivery systems: from laboratory to clinic: a review of the recently introduced commercial liposomal and lipid based formulations of amphotericin B. Adv. Drug Deliv. Rev. 1997;24:345–363. doi: 10.1016/S0169-409X(96)00496-6. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

