Antibodies to envelope glycoprotein of dengue virus during the natural course of infection are predominantly cross-reactive and recognize epitopes containing highly conserved residues at the fusion loop of domain II
- PMID: 18448542
- PMCID: PMC2447043
- DOI: 10.1128/JVI.00316-08
Antibodies to envelope glycoprotein of dengue virus during the natural course of infection are predominantly cross-reactive and recognize epitopes containing highly conserved residues at the fusion loop of domain II
Abstract
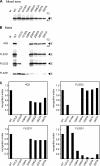
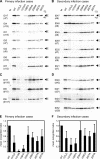
The antibody response to the envelope (E) glycoprotein of dengue virus (DENV) is known to play a critical role in both protection from and enhancement of disease, especially after primary infection. However, the relative amounts of homologous and heterologous anti-E antibodies and their epitopes remain unclear. In this study, we examined the antibody responses to E protein as well as to precursor membrane (PrM), capsid, and nonstructural protein 1 (NS1) of four serotypes of DENV by Western blot analysis of DENV serotype 2-infected patients with different disease severity and immune status during an outbreak in southern Taiwan in 2002. Based on the early-convalescent-phase sera tested, the rates of antibody responses to PrM and NS1 proteins were significantly higher in patients with secondary infection than in those with primary infection. A blocking experiment and neutralization assay showed that more than 90% of anti-E antibodies after primary infection were cross-reactive and nonneutralizing against heterologous serotypes and that only a minor proportion were type specific, which may account for the type-specific neutralization activity. Moreover, the E-binding activity in sera of 10 patients with primary infection was greatly reduced by amino acid replacements of three fusion loop residues, tryptophan at position 101, leucine at position 107, and phenylalanine at position 108, but not by replacements of those outside the fusion loop of domain II, suggesting that the predominantly cross-reactive anti-E antibodies recognized epitopes involving the highly conserved residues at the fusion loop of domain II. These findings have implications for our understanding of the pathogenesis of dengue and for the future design of subunit vaccine against DENV as well.
Figures




References
-
- Alvarez, M., R. Rodriguez-Roche, L. Bernardo, S. Vazquez, L. Morier, D. Gonzalez, O. Castro, G. Kouri, S. B. Halstead, and M. G. Guzman. 2006. Dengue hemorrhagic Fever caused by sequential dengue 1-3 virus infections over a long time interval: Havana epidemic, 2001-2002. Am. J. Trop. Med. Hyg. 751113-1117. - PubMed
-
- Bray, M., and C. J. Lai. 1991. Dengue virus premembrane and membrane proteins elicit a protective immune response. Virology 185505-508. - PubMed
-
- Burke, D. S., A. Nisalak, D. E. Johnson, and R. M. Scott. 1988. A prospective study of dengue infections in Bangkok. Am. J. Trop. Med. Hyg. 38172-180. - PubMed
-
- Churdboonchart, V., N. Bhamarapravati, S. Peampramprecha, and S. Sirinavin. 1991. Antibodies against dengue viral proteins in primary and secondary dengue hemorrhagic fever. Am. J. Trop. Med. Hyg. 44481-493. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

