Regulation of epithelial sodium transport by promyelocytic leukemia zinc finger protein
- PMID: 18448589
- PMCID: PMC2494520
- DOI: 10.1152/ajprenal.00573.2007
Regulation of epithelial sodium transport by promyelocytic leukemia zinc finger protein
Abstract
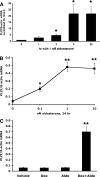
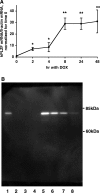
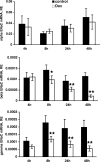
Aldosterone is the principal regulator of Na homeostasis, and thereby blood pressure. One of the main targets of aldosterone is the epithelial Na channel (ENaC) located in the apical membrane of target cells. Previous studies identified several genes involved in the regulation of ENaC such as SGK1; however, SGK1 knockout mice have only a mild salt-losing phenotype, indicating that further genes must be involved in the action of aldosterone. In our search for further aldosterone-regulated genes, we discovered that aldosterone, at physiological concentrations, induces the expression of the promyelocytic leukemia zinc finger protein (PLZF) in renal cortical collecting duct (CCD) cell lines that stably express mineralocorticoid receptors (MRs). This effect is rapid and does not require de novo protein synthesis, suggesting a direct action. Surprisingly, stable overexpression of human or mouse PLZF isoforms significantly decreased transepithelial Na transport in CCD cells while having no effect on the integrity of the monolayers. In parallel with the decline in Na transport, PLZF suppressed the mRNA levels of beta- and gamma-ENaC subunits. These observations suggest that PLZF is a negative regulator of ENaC in renal epithelial cells and might be part of a negative feedback loop that limits aldosterone's stimulatory effects on sodium reabsorption.
Figures
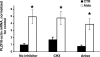




Similar articles
-
Regulation of sodium transport in mammalian collecting duct cells by aldosterone-induced kinase, SGK1: structure/function studies.Mol Cell Endocrinol. 2004 Mar 31;217(1-2):197-202. doi: 10.1016/j.mce.2003.10.043. Mol Cell Endocrinol. 2004. PMID: 15134818
-
Aldosterone-dependent and -independent regulation of the epithelial sodium channel (ENaC) in mouse distal nephron.Am J Physiol Renal Physiol. 2012 Nov 1;303(9):F1289-99. doi: 10.1152/ajprenal.00247.2012. Epub 2012 Aug 29. Am J Physiol Renal Physiol. 2012. PMID: 22933298
-
Protein kinase D2 regulates epithelial sodium channel activity and aldosterone non-genomic responses in renal cortical collecting duct cells.Steroids. 2020 Mar;155:108553. doi: 10.1016/j.steroids.2019.108553. Epub 2019 Dec 11. Steroids. 2020. PMID: 31836481
-
The sgk, an aldosterone-induced gene in mineralocorticoid target cells, regulates the epithelial sodium channel.Kidney Int. 2000 Apr;57(4):1290-4. doi: 10.1046/j.1523-1755.2000.00964.x. Kidney Int. 2000. PMID: 10760056 Review.
-
Short-term aldosterone action on Na,K-ATPase surface expression: role of aldosterone-induced SGK1?Ann N Y Acad Sci. 2003 Apr;986:554-61. doi: 10.1111/j.1749-6632.2003.tb07253.x. Ann N Y Acad Sci. 2003. PMID: 12763889 Review.
Cited by
-
The physiological impact of the serum and glucocorticoid-inducible kinase SGK1.Curr Opin Nephrol Hypertens. 2009 Sep;18(5):439-48. doi: 10.1097/MNH.0b013e32832f125e. Curr Opin Nephrol Hypertens. 2009. PMID: 19584721 Free PMC article. Review.
-
Aldosterone: Renal Action and Physiological Effects.Compr Physiol. 2023 Mar 30;13(2):4409-4491. doi: 10.1002/cphy.c190043. Compr Physiol. 2023. PMID: 36994769 Free PMC article.
-
Establishment of Murine Pregnancy Requires the Promyelocytic Leukemia Zinc Finger Transcription Factor.Int J Mol Sci. 2024 Mar 19;25(6):3451. doi: 10.3390/ijms25063451. Int J Mol Sci. 2024. PMID: 38542422 Free PMC article.
-
Bioinformatics Analysis of ZBTB16 as a Prognostic Marker for Ewing's Sarcoma.Biomed Res Int. 2021 Oct 6;2021:1989917. doi: 10.1155/2021/1989917. eCollection 2021. Biomed Res Int. 2021. PMID: 34660783 Free PMC article.
-
Regulation of sodium transport by ENaC in the kidney.Curr Opin Nephrol Hypertens. 2010 Jan;19(1):98-105. doi: 10.1097/MNH.0b013e328332bda4. Curr Opin Nephrol Hypertens. 2010. PMID: 19996890 Free PMC article. Review.
References
-
- Alvarez de la Rosa D, Canessa CM. Role of SGK in hormonal regulation of epithelial sodium channel in A6 cells. Am J Physiol Cell Physiol 284: C404–C414, 2003. - PubMed
-
- Barna M, Hawe N, Niswander L, Pandolfi PP. Plzf regulates limb and axial skeletal patterning. Nat Genet 25: 166–172, 2000. - PubMed
-
- Boyd C, Naray-Fejes-Tóth A. Gene regulation of ENaC subunits by serum- and glucocorticoid-inducible kinase-1. Am J Physiol Renal Physiol 288: F505–F512, 2005. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

