Structural insights into aberrant topological patterns of large-scale cortical networks in Alzheimer's disease
- PMID: 18448652
- PMCID: PMC6670444
- DOI: 10.1523/JNEUROSCI.0141-08.2008
Structural insights into aberrant topological patterns of large-scale cortical networks in Alzheimer's disease
Abstract
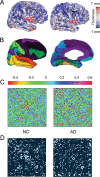
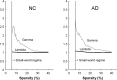
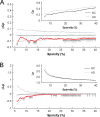
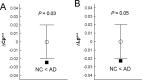
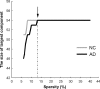
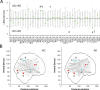
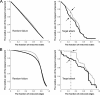
Recent research on Alzheimer's disease (AD) has shown that cognitive and memory decline in this disease is accompanied by disrupted changes in the coordination of large-scale brain functional networks. However, alterations in coordinated patterns of structural brain networks in AD are still poorly understood. Here, we used cortical thickness measurement from magnetic resonance imaging to investigate large-scale structural brain networks in 92 AD patients and 97 normal controls. Brain networks were constructed by thresholding cortical thickness correlation matrices of 54 regions and analyzed using graph theoretical approaches. Compared with controls, AD patients showed decreased cortical thickness intercorrelations between the bilateral parietal regions and increased intercorrelations in several selective regions involving the lateral temporal and parietal cortex as well as the cingulate and medial frontal cortex regions. Specially, AD patients showed abnormal small-world architecture in the structural cortical networks (increased clustering and shortest paths linking individual regions), implying a less optimal topological organization in AD. Moreover, AD patients were associated with reduced nodal centrality predominantly in the temporal and parietal heteromodal association cortex regions and increased nodal centrality in the occipital cortex regions. Finally, the brain networks of AD were about equally as robust to random failures as those of controls, but more vulnerable against targeted attacks, presumably because of the effects of pathological topological organization. Our findings suggest that the coordinated patterns of cortical morphology are widely altered in AD patients, thus providing structural evidence for disrupted integrity in large-scale brain networks that underlie cognition. This work has implications for our understanding of how functional deficits in patients are associated with their underlying structural (morphological) basis.
Figures
Comment in
-
Alzheimer's disease: a search for broken links.J Neurosci. 2008 Aug 13;28(33):8148-9. doi: 10.1523/JNEUROSCI.2433-08.2008. J Neurosci. 2008. PMID: 18701676 Free PMC article. Review. No abstract available.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
