Distinct cellular and molecular mechanisms underlie functional remodeling of repolarizing K+ currents with left ventricular hypertrophy
- PMID: 18451341
- PMCID: PMC2653713
- DOI: 10.1161/CIRCRESAHA.107.170050
Distinct cellular and molecular mechanisms underlie functional remodeling of repolarizing K+ currents with left ventricular hypertrophy
Abstract
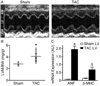
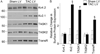
Left ventricular hypertrophy (LVH) is associated with electric remodeling and increased arrhythmia risk, although the underlying mechanisms are poorly understood. In the experiments here, functional voltage-gated (Kv) and inwardly rectifying (Kir) K(+) channel remodeling was examined in a mouse model of pressure overload-induced LVH, produced by transverse aortic constriction (TAC). Action potential durations (APDs) at 90% repolarization in TAC LV myocytes and QT(c) intervals in TAC mice were prolonged. Mean whole-cell membrane capacitance (C(m)) was higher, and I(to,f), I(K,slow), I(ss), and I(K1) densities were lower in TAC, than in sham, LV myocytes. Although the primary determinant of the reduced current densities is the increase in C(m), I(K,slow) amplitudes were decreased and I(ss) amplitudes were increased in TAC LV cells. Further experiments revealed regional differences in the effects of LVH. Cellular hypertrophy and increased I(ss) amplitudes were more pronounced in TAC endocardial LV cells, whereas I(K,slow) amplitudes were selectively reduced in TAC epicardial LV cells. Consistent with the similarities in I(to,f) and I(K1) amplitudes, Kv4.2, Kv4.3, and KChIP2 (I(to,f)), as well as Kir2.1 and Kir2.2 (I(K1)), transcript and protein expression levels were similar in TAC and sham LV. Unexpectedly, expression of I(K,slow) channel subunits Kv1.5 and Kv2.1 was increased in TAC LV. Biochemical experiments also demonstrated that, although total protein was unaltered, cell surface expression of TASK1 was increased in TAC LV. Functional changes in repolarizing K(+) currents with LVH, therefore, result from distinct cellular (cardiomyocyte enlargement) and molecular (alterations in the numbers of functional channels) mechanisms.
Figures





Comment in
-
Effects of heart disease on cardiac ion current density versus current amplitude: important conceptual subtleties in the language of arrhythmogenic ion channel remodeling.Circ Res. 2008 Jun 6;102(11):1298-300. doi: 10.1161/CIRCRESAHA.108.178087. Circ Res. 2008. PMID: 18535265 No abstract available.
References
-
- Schoen FJ. The heart. In: Cotran RS, Kumar V, Collins T, editors. Robbin’s Pathologic Basis of Disease. Philadelphia, Pa: WB Saunders Co; 1999. pp. 543–599.
-
- McMullen JR, Jennings GL. Differences between pathological and physiological cardiac hypertrophy: novel therapeutic strategies to treat heart failure. Clin Exp Pharmacol Physiol. 2007;34:255–262. - PubMed
-
- Nattel S, Maguy A, Le Bouter S, Yeh YH. Arrhythmogenic ion-channel remodeling in the heart: heart failure, myocardial infarction, and atrial fibrillation. Physiol Rev. 2007;87:425–456. - PubMed
-
- Armoundas AA, Wu R, Juang G, Marban E, Tomaselli GF. Electrical and structural remodeling of the failing ventricle. Pharmacol Ther. 2001;92:213–230. - PubMed
-
- Kaab S, Nuss HB, Chiamvimonvat N, O’Rourke B, Pak PH, Kass DA, Marban E, Tomaselli GF. Ionic mechanism of action potential prolongation in ventricular myocytes from dogs with pacing-induced heart failure. Circ Res. 1996;78:262–273. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

