Modulation of self-association and subsequent fibril formation in an alanine-rich helical polypeptide
- PMID: 18452331
- PMCID: PMC2606058
- DOI: 10.1021/bm800056r
Modulation of self-association and subsequent fibril formation in an alanine-rich helical polypeptide
Abstract
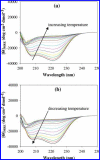
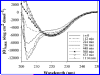
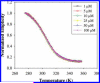
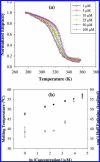
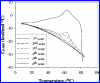
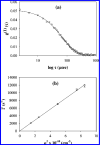
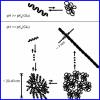
Thermal unfolding, reversible self-association, and irreversible aggregation were investigated for an alanine-rich helical polypeptide, 17-H-6, with sequence [AAAQEAAAAQAAAQAEAAQAAQ] 6. Dynamic light scattering, transmission electron microscopy, and thermal unfolding measurements indicate that 17-H-6 spontaneously and reversibly self-associates at acidic pH and low temperature. The resulting multimers have a compact, globular morphology with an average hydrodynamic radius approximately 10-20 nm and reversibly dissociate to monomers upon an increase to pH 7.4. Both free monomer and 17-H-6 chains within the multimers are alpha-helical and folded at low temperature. Reversible unfolding of the monomer occurs upon heating of solutions at pH 7.4. At pH 2.3, heating first causes incomplete dissociation and unfolding of the constituent chains. Further incubation at elevated temperature induces additional structural and morphological changes and results in fibrils with a beta-sheet 2 degrees structure and a characteristic diameter of 5-10 nm (7 nm mean). The ability to modulate association and aggregation suggests opportunities for this class of polypeptides in nanotechnology and biomedical applications.
Figures




Similar articles
-
Conformational and aggregation properties of a PEGylated alanine-rich polypeptide.Biomacromolecules. 2011 Jun 13;12(6):2184-92. doi: 10.1021/bm200272w. Epub 2011 May 9. Biomacromolecules. 2011. PMID: 21553871 Free PMC article.
-
Characterizing aggregate growth and morphology of alanine-rich polypeptides as a function of sequence chemistry and solution temperature from scattering, spectroscopy, and microscopy.Biophys Chem. 2020 Dec;267:106481. doi: 10.1016/j.bpc.2020.106481. Epub 2020 Sep 25. Biophys Chem. 2020. PMID: 33035751 Free PMC article.
-
Biophysical investigations on the aggregation and thermal unfolding of harpin(Pss) and identification of leucine-zipper-like motifs in harpins.Biochim Biophys Acta. 2009 Nov;1794(11):1684-92. doi: 10.1016/j.bbapap.2009.07.023. Epub 2009 Aug 7. Biochim Biophys Acta. 2009. PMID: 19665594
-
Stability, unfolding, and structural changes of cofactor-free 1H-3-hydroxy-4-oxoquinaldine 2,4-dioxygenase.Biochemistry. 2007 Apr 10;46(14):4241-9. doi: 10.1021/bi0622423. Epub 2007 Mar 20. Biochemistry. 2007. PMID: 17371045
-
Multivalent protein polymers with controlled chemical and physical properties.Adv Drug Deliv Rev. 2010 Dec 30;62(15):1530-40. doi: 10.1016/j.addr.2010.05.002. Epub 2010 Jun 1. Adv Drug Deliv Rev. 2010. PMID: 20562016 Free PMC article. Review.
Cited by
-
Architecture effects on L-selectin shedding induced by polypeptide-based multivalent ligands.Polym Chem. 2011 Jul;2(7):1513-1522. doi: 10.1039/C1PY00063B. Polym Chem. 2011. PMID: 23926449 Free PMC article.
-
Assembly Properties of an Alanine-Rich, Lysine-Containing Peptide and the Formation of Peptide/Polymer Hybrid Hydrogels.Macromol Chem Phys. 2011 Feb 1;212(3):229-239. doi: 10.1002/macp.201000446. Macromol Chem Phys. 2011. PMID: 21359141 Free PMC article.
-
Engineering structure and function using thermoresponsive biopolymers.Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2016 Jan-Feb;8(1):123-38. doi: 10.1002/wnan.1350. Epub 2015 Jun 26. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2016. PMID: 26112277 Free PMC article. Review.
-
Conformational and aggregation properties of a PEGylated alanine-rich polypeptide.Biomacromolecules. 2011 Jun 13;12(6):2184-92. doi: 10.1021/bm200272w. Epub 2011 May 9. Biomacromolecules. 2011. PMID: 21553871 Free PMC article.
-
Protein- and peptide-modified synthetic polymeric biomaterials.Biopolymers. 2010;94(1):32-48. doi: 10.1002/bip.21333. Biopolymers. 2010. PMID: 20091878 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

