National Emphysema Treatment Trial: the major outcomes of lung volume reduction surgery in severe emphysema
- PMID: 18453345
- PMCID: PMC2645309
- DOI: 10.1513/pats.200801-013ET
National Emphysema Treatment Trial: the major outcomes of lung volume reduction surgery in severe emphysema
Abstract
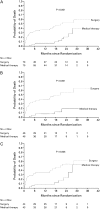
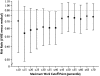
The National Emphysema Treatment Trial (NETT) has published many articles reporting the various outcomes of lung volume reduction surgery versus medical treatment for patients with severe emphysema. However, long and complex clinical trials like NETT that involve both medical and surgical issues generate multiple manuscripts over a period of years and report an array of various outcomes. As a result, the essential findings of the trial may appear to be fragmented to the clinician or clinical researcher or be lost among the many medical reports published each year. In this review, we summarize in one publication the major medical and surgical outcomes of NETT.
Figures

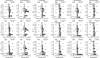


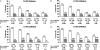
References
-
- National Emphysema Treatment Trial Research Group. A randomized trial comparing lung-volume-reduction surgery with medical therapy for severe emphysema. N Engl J Med 2003;348:2059–2073. - PubMed
-
- Naunheim KS, Wood DE, Mohsenifar Z, Sternberg AL, Criner GJ, DeCamp MM, Deschamps CC, Martinez FJ, Sciurba FC, Tonascia J, et al. Long-term follow-up of patients receiving lung-volume-reduction surgery versus medical therapy for severe emphysema. Ann Thorac Surg 2006;82:431–443. - PubMed
-
- DeCamp MM, Blackstone EH, Naunheim KS, Krasna MJ, Wood DE, Meli YM, McKenna RJ; for the NETT Research Group. Patient and surgical factors influencing air leak after lung volume reduction surgery: lessons learned from the National Emphysema Treatment Trial. Ann Thorac Surg 2006;82:197–207. - PubMed
-
- Naunheim KS, Wood, DE, Krasna MJ, Decamp MM, Ginsburg ME, McKenna RJ, Criner GJ, Hoffman EA, Sternberg AL, Deschamps CC; for the National Emphysema Treatment Trial Research Group. Predictors of operative mortality and cardiopulmonary morbidity in the National Emphysema Treatment Trial. J Thorac Cardiovasc Surg 2006;131:43–53. - PubMed
-
- National Emphysema Treatment Trial Research Group. Safety and efficacy of median sternotomy versus video-assisted thoracic surgery for lung volume reduction surgery. J Thorac Cardiovasc Surg 2004;127:1350–1360. - PubMed
Publication types
MeSH terms
Grants and funding
- N01HR76113/HR/NHLBI NIH HHS/United States
- N01HR76107/HR/NHLBI NIH HHS/United States
- N01HR76102/HR/NHLBI NIH HHS/United States
- N01HR76104/HR/NHLBI NIH HHS/United States
- N01HR76118/HR/NHLBI NIH HHS/United States
- N01HR76115/HR/NHLBI NIH HHS/United States
- N01HR76105/HR/NHLBI NIH HHS/United States
- N01HR76111/HR/NHLBI NIH HHS/United States
- N01HR76114/HR/NHLBI NIH HHS/United States
- N01HR76103/HR/NHLBI NIH HHS/United States
- N01HR76110/HR/NHLBI NIH HHS/United States
- N01HR76119/HR/NHLBI NIH HHS/United States
- N01HR76108/HR/NHLBI NIH HHS/United States
- N01HR76106/HR/NHLBI NIH HHS/United States
- N01HR76109/HR/NHLBI NIH HHS/United States
- N01HR76116/HR/NHLBI NIH HHS/United States
- N01HR76112/HR/NHLBI NIH HHS/United States
- N01HR76101/HR/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Research Materials
