Blockade of virus infection by human CD4+ T cells via a cytokine relay network
- PMID: 18453613
- PMCID: PMC2387070
- DOI: 10.4049/jimmunol.180.10.6923
Blockade of virus infection by human CD4+ T cells via a cytokine relay network
Abstract
CD4(+) T cells directly participate in bacterial clearance through secretion of proinflammatory cytokines. Although viral clearance relies heavily on CD8(+) T cell functions, we sought to determine whether human CD4(+) T cells could also directly influence viral clearance through cytokine secretion. We found that IFN-gamma and TNF-alpha, secreted by IL-12-polarized Th1 cells, displayed potent antiviral effects against a variety of viruses. IFN-gamma and TNF-alpha acted directly to inhibit hepatitis C virus replication in an in vitro replicon system, and neutralization of both cytokines was required to block the antiviral activity that was secreted by Th1 cells. IFN-gamma and TNF-alpha also exerted antiviral effects against vesicular stomatitis virus infection, but in this case, functional type I IFN receptor activity was required. Thus, in cases of vesicular stomatitis virus infection, the combination of IFN-gamma and TNF-alpha secreted by human Th1 cells acted indirectly through the IFN-alpha/beta receptor. These results highlight the importance of CD4(+) T cells in directly regulating antiviral responses through proinflammatory cytokines acting in both a direct and indirect manner.
Figures
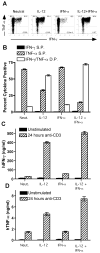
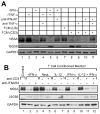
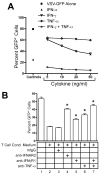
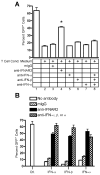
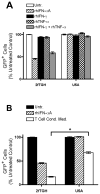
 ), rhTNF-α (◇), or rhIFN-γ + rhTNF-α (○) as indicated. (B) THP-1 cells were infected in the absence (1) or presence (–7) of 10% (v/v) T cell conditioned media from IL-12 + IFN-α activated T cells in the absence (2) or presence of 5 μg/ml mouse IgG1 isotype control antibody (3), 5 μg/ml anti-hIFNAR2 (4), 10 μg/ml anti-hIFNγR1 (5), 5 μg/ml anti-hTNF-α (6), or a combination of anti-hIFNγR1 and anti-hTNF-α (7).
), rhTNF-α (◇), or rhIFN-γ + rhTNF-α (○) as indicated. (B) THP-1 cells were infected in the absence (1) or presence (–7) of 10% (v/v) T cell conditioned media from IL-12 + IFN-α activated T cells in the absence (2) or presence of 5 μg/ml mouse IgG1 isotype control antibody (3), 5 μg/ml anti-hIFNAR2 (4), 10 μg/ml anti-hIFNγR1 (5), 5 μg/ml anti-hTNF-α (6), or a combination of anti-hIFNγR1 and anti-hTNF-α (7).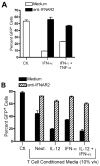
Similar articles
-
IL-12 is not required for induction of type 1 cytokine responses in viral infections.J Immunol. 1999 Jan 15;162(2):965-73. J Immunol. 1999. PMID: 9916721
-
T helper 1/T helper 2 cytokine imbalance in respiratory syncytial virus infection is associated with increased endogenous plasma cortisol.Pediatrics. 2006 May;117(5):e878-86. doi: 10.1542/peds.2005-2119. Epub 2006 Apr 17. Pediatrics. 2006. PMID: 16618789
-
Interleukin 7 stimulates tumour necrosis factor alpha and Th1 cytokine production in joints of patients with rheumatoid arthritis.Ann Rheum Dis. 2003 Feb;62(2):113-9. doi: 10.1136/ard.62.2.113. Ann Rheum Dis. 2003. PMID: 12525379 Free PMC article.
-
IL-12 producing monocytes and IFN-gamma and TNF-alpha producing T-lymphocytes are increased in placentas infected by Plasmodium falciparum.J Reprod Immunol. 2007 Jun;74(1-2):152-62. doi: 10.1016/j.jri.2006.10.001. Epub 2006 Dec 27. J Reprod Immunol. 2007. PMID: 17194481
-
Stability of Regulatory T Cells Undermined or Endorsed by Different Type-1 Cytokines.Adv Exp Med Biol. 2015;850:17-30. doi: 10.1007/978-3-319-15774-0_2. Adv Exp Med Biol. 2015. PMID: 26324343 Review.
Cited by
-
Turning the tables on cytomegalovirus: targeting viral Fc receptors by CARs containing mutated CH2-CH3 IgG spacer domains.J Transl Med. 2018 Feb 8;16(1):26. doi: 10.1186/s12967-018-1394-x. J Transl Med. 2018. PMID: 29422056 Free PMC article.
-
Cutting edge: a T-bet-independent role for IFN-alpha/beta in regulating IL-2 secretion in human CD4+ central memory T cells.J Immunol. 2008 Dec 15;181(12):8204-8. doi: 10.4049/jimmunol.181.12.8204. J Immunol. 2008. PMID: 19050236 Free PMC article.
-
Phytochemicals and Immunomodulatory Effect of Nelumbo nucifera Flower Extracts on Human Macrophages.Plants (Basel). 2021 Sep 24;10(10):2007. doi: 10.3390/plants10102007. Plants (Basel). 2021. PMID: 34685815 Free PMC article.
-
Involvement of activation of PKR in HBx-siRNA-mediated innate immune effects on HBV inhibition.PLoS One. 2011;6(12):e27931. doi: 10.1371/journal.pone.0027931. Epub 2011 Dec 8. PLoS One. 2011. PMID: 22174754 Free PMC article.
-
A gB/CD3 bispecific BiTE antibody construct for targeting Human Cytomegalovirus-infected cells.Sci Rep. 2018 Nov 28;8(1):17453. doi: 10.1038/s41598-018-36055-2. Sci Rep. 2018. PMID: 30487534 Free PMC article.
References
-
- Canessa A, Pistoia V, Roncella S, Merli A, Melioli G, Terragna A, Ferrarini M. An in vitro model for Toxoplasma infection in man: interaction between CD4+ monoclonal T cells and macrophages results in killing of trophozoites. J Immunol. 1988;140:3580–3588. - PubMed
-
- Geginat G, Lalic M, Kretschmar M, Goebel W, Hof H, Palm D, Bubert A. Th1 cells specific for a secreted protein of Listeria monocytogenes are protective in vivo. J Immunol. 1998;160:6046–6055. - PubMed
-
- Seder RA, Ahmed R. Similarities and differences in CD4+ and CD8+ effector and memory T cell generation. Nat Immunol. 2003;4:835–842. - PubMed
-
- Urbani S, Amadei B, Fisicaro P, Tola D, Orlandini A, Sacchelli L, Mori C, Missale G, Ferrari C. Outcome of acute hepatitis C is related to virus-specific CD4 function and maturation of antiviral memory CD8 responses. Hepatology. 2006;44:126–139. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

