The hepatic stem cell niche: identification by label-retaining cell assay
- PMID: 18454509
- PMCID: PMC2847183
- DOI: 10.1002/hep.22218
The hepatic stem cell niche: identification by label-retaining cell assay
Abstract
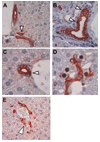
Label retention assays remain the state-of-the-art approach to identify the location of intraorgan epithelial stem cell niches, in situ and in vivo. They are commonly used in organs with rapid cell turnover but have not been applied to the liver, where cell turnover is very slow. We used a sublethal dose of acetaminophen administered coincident with bromodeoxyuridine to load possible hepatic stem cells in mice with label and then administered a second, sublethal chase of acetaminophen to accomplish "washout" of label from transit amplifying cell populations.
Conclusion: Four possible hepatic stem cell niches are identified by this approach: the canal of Hering (proximal biliary tree), intralobular bile ducts, periductal "null" mononuclear cells, and peribiliary hepatocytes. These results confirm several different and often contradictory lines of investigation regarding the intrahepatic location of stem/progenitor cells and suggest that the liver has a multi-tiered, flexible system of regeneration rather than a single stem/progenitor cell location.
Conflict of interest statement
Potential conflict of interest: Nothing to report.
Figures




Comment in
-
Location is everything: the liver stem cell niche.Hepatology. 2008 Jun;47(6):1810-2. doi: 10.1002/hep.22333. Hepatology. 2008. PMID: 18506878 No abstract available.
References
-
- Sell S. Heterogeneity and plasticity of hepatocyte lineage cells. HEPATOLOGY. 2001;33:738–750. - PubMed
-
- Rabes HM, Wirshing R, Ruxzek HV, Iseler G. Analysis of cell cycle compartments of hepatocytes after partial hepatectomy. Cell Tissue Kinet. 1976;9:517–532. - PubMed
-
- Grisham JW, Thorgeirsson SS. Liver stem cells. In: Potten CS, editor. Stem cells. New York: Academic Press; 1997. pp. 233–282.
-
- Forbes S, Vig P, Poulsom R, Thomas H, Alison M. Hepatic stem cells. J Pathol. 2002;197:510–518. - PubMed
-
- Newsome PN, Hussain MA, Theise ND. Hepatic oval cells: helping redefine a paradigm in stem cell biology. Curr Top Dev Biol. 2004;61:1–28. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
