Regulation of in situ to invasive breast carcinoma transition
- PMID: 18455123
- PMCID: PMC3705908
- DOI: 10.1016/j.ccr.2008.03.007
Regulation of in situ to invasive breast carcinoma transition
Abstract
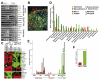
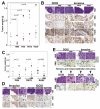
The transition of ductal carcinoma in situ (DCIS) to invasive carcinoma is a poorly understood key event in breast tumor progression. Here, we analyzed the role of myoepithelial cells and fibroblasts in the progression of in situ carcinomas using a model of human DCIS and primary breast tumors. Progression to invasion was promoted by fibroblasts and inhibited by normal myoepithelial cells. Molecular profiles of isolated luminal epithelial and myoepithelial cells identified an intricate interaction network involving TGFbeta, Hedgehog, cell adhesion, and p63 required for myoepithelial cell differentiation, the elimination of which resulted in loss of myoepithelial cells and progression to invasion.
Figures



References
-
- Allinen M, Beroukhim R, Cai L, Brennan C, Lahti-Domenici J, Huang H, Porter D, Hu M, Chin L, Richardson A, et al. Molecular characterization of the tumor microenvironment in breast cancer. Cancer Cell. 2004;6:17–32. - PubMed
-
- Allred DC, Mohsin SK, Fuqua SA. Histological and biological evolution of human premalignant breast disease. Endocr. Relat. Cancer. 2001;8:47–61. - PubMed
-
- Barsky SH, Karlin NJ. Myoepithelial cells: Autocrine and paracrine suppressors of breast cancer progression. J. Mammary Gland Biol. Neoplasia. 2005;10:249–260. - PubMed
-
- Burstein HJ, Polyak K, Wong JS, Lester SC, Kaelin CM. Ductal carcinoma in situ of the breast. N. Engl. J. Med. 2004;350:1430–1441. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

