Salivary progesterone and estriol among pregnant women treated with 17-alpha-hydroxyprogesterone caproate or placebo
- PMID: 18456237
- PMCID: PMC2794481
- DOI: 10.1016/j.ajog.2008.03.003
Salivary progesterone and estriol among pregnant women treated with 17-alpha-hydroxyprogesterone caproate or placebo
Abstract
Objective: The objectives of the study was to determine whether salivary progesterone (P) or estriol (E3) concentration at 16-20 weeks' gestation predicts preterm birth or the response to 17alpha-hydroxyprogesterone caproate (17OHPC) and whether 17OHPC treatment affected the trajectory of salivary P and E3 as pregnancy progressed.
Study design: This was a secondary analysis of a clinical trial of 17OHPC to prevent preterm birth. Baseline saliva was assayed for P and E3. Weekly salivary samples were obtained from 40 women who received 17OHPC and 40 who received placebo in a multicenter randomized trial of 17OHPC to prevent recurrent preterm delivery.
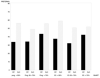
Results: Both low and high baseline saliva P and E3 were associated with a slightly increased risk of preterm birth. However, 17OHPC prevented preterm birth comparably, regardless of baseline salivary hormone concentrations. 17OHPC did not alter the trajectory of salivary P over pregnancy, but it significantly blunted the rise in salivary E3 as well as the rise in the E3/P ratio.
Conclusion: 17OHPC flattened the trajectory of E3 in the second half of pregnancy, suggesting that the drug influences the fetoplacental unit.
Figures

References
-
- Csapo AI. Progesterone “block.”. Am J Anat. 1956;98:273–91. - PubMed
-
- Cousins LM, Hobel CJ, Chang RJ, Okada DM, Marshall JR. Serum progesterone and estradiol-17b levels in premature and term labor. Am J Obstet Gynecol. 1977;127:612–5. - PubMed
-
- Johnson JWC, Lee PA, Zachary AS, Calhoun S, Migeon CJ. High-risk prematurity—progestin treatment and steroid studies. Obstet Gynecol. 1979;54:412–8. - PubMed
-
- Block BS, Liggins GC, Creasy RK. Preterm delivery is not predicted by serial plasma estradiol or progesterone concentration measurements. Am J Obstet Gynecol. 1984;150:716–22. - PubMed
-
- Goldman S, Weiss A, Altmalah I, Shalev E. Progesterone receptor in human decidua and fetal membranes before and after contractions: possible mechanism for functional progesterone withdrawal. Mol Hum Reprod. 2005;11:269–77. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- HD36801/HD/NICHD NIH HHS/United States
- UG1 HD027869/HD/NICHD NIH HHS/United States
- U10 HD040544/HD/NICHD NIH HHS/United States
- U10 HD034136/HD/NICHD NIH HHS/United States
- U10 HD040485/HD/NICHD NIH HHS/United States
- U10 HD034116/HD/NICHD NIH HHS/United States
- HD40560/HD/NICHD NIH HHS/United States
- U10 HD027915/HD/NICHD NIH HHS/United States
- U10 HD040560/HD/NICHD NIH HHS/United States
- U10 HD040512/HD/NICHD NIH HHS/United States
- HD40544/HD/NICHD NIH HHS/United States
- U01 HD036801/HD/NICHD NIH HHS/United States
- U10 HD040500/HD/NICHD NIH HHS/United States
- HD27861/HD/NICHD NIH HHS/United States
- UG1 HD034116/HD/NICHD NIH HHS/United States
- HD27869/HD/NICHD NIH HHS/United States
- HD34136/HD/NICHD NIH HHS/United States
- UG1 HD040560/HD/NICHD NIH HHS/United States
- HD27860/HD/NICHD NIH HHS/United States
- UG1 HD027915/HD/NICHD NIH HHS/United States
- HD21414/HD/NICHD NIH HHS/United States
- U10 HD027905/HD/NICHD NIH HHS/United States
- HD40512/HD/NICHD NIH HHS/United States
- UG1 HD040544/HD/NICHD NIH HHS/United States
- UG1 HD034208/HD/NICHD NIH HHS/United States
- UG1 HD040512/HD/NICHD NIH HHS/United States
- HD34210/HD/NICHD NIH HHS/United States
- HD21410/HD/NICHD NIH HHS/United States
- U10 HD027869/HD/NICHD NIH HHS/United States
- U10 HD027917/HD/NICHD NIH HHS/United States
- HD34116/HD/NICHD NIH HHS/United States
- U10 HD034122/HD/NICHD NIH HHS/United States
- U10 HD027860/HD/NICHD NIH HHS/United States
- HD40500/HD/NICHD NIH HHS/United States
- U10 HD034208/HD/NICHD NIH HHS/United States
- HD34208/HD/NICHD NIH HHS/United States
- HD27915/HD/NICHD NIH HHS/United States
- UG1 HD040500/HD/NICHD NIH HHS/United States
- HD27917/HD/NICHD NIH HHS/United States
- NIH0010045861/ImNIH/Intramural NIH HHS/United States
- U10 HD021410/HD/NICHD NIH HHS/United States
- U10 HD036801/HD/NICHD NIH HHS/United States
- U24 HD036801/HD/NICHD NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

