The fe2+ site of photosynthetic reaction centers probed by multiple scattering x-ray absorption fine structure spectroscopy: improving structure resolution in dry matrices
- PMID: 18456824
- PMCID: PMC2440445
- DOI: 10.1529/biophysj.108.132654
The fe2+ site of photosynthetic reaction centers probed by multiple scattering x-ray absorption fine structure spectroscopy: improving structure resolution in dry matrices
Abstract
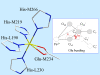
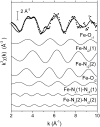
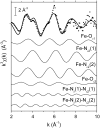
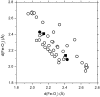
We report on the x-ray absorption fine structure of the Fe(2+) site in photosynthetic reaction centers from Rhodobacter sphaeroides. Crystallographic studies show that Fe(2+) is ligated with four N(epsilon) atoms from four histidine (His) residues and two O(epsilon) atoms from a Glu residue. By considering multiple scattering contributions to the x-ray absorption fine structure function, we improved the structural resolution of the site: His residues were split into two groups, characterized by different Fe-N(epsilon) distances, and two distinct Fe-O(epsilon) bond lengths resolved. The effect of the environment was studied by embedding the reaction centers into a polyvinyl alcohol film and into a dehydrated trehalose matrix. Incorporation into trehalose caused elongation in one of the two Fe-N(epsilon) distances, and in one Fe-O(epsilon) bond length, compared with the polyvinyl alcohol film. The asymmetry detected in the cluster of His residues and its response to incorporation into trehalose are ascribed to the hydrogen bonds between two His residues and the quinone acceptors. The structural distortions observed in the trehalose matrix indicate a strong interaction between the reaction-centers surface and the water-trehalose matrix, which propagates deeply into the interior of the protein. The absence of matrix effects on the Debye-Waller factors is brought back to the static heterogeneity and rigidity of the ligand cluster.
Figures
Similar articles
-
Multiple scattering x-ray absorption studies of Zn2+ binding sites in bacterial photosynthetic reaction centers.Biophys J. 2005 Mar;88(3):2038-46. doi: 10.1529/biophysj.104.050971. Epub 2004 Dec 21. Biophys J. 2005. PMID: 15613631 Free PMC article.
-
Cytochrome C in a dry trehalose matrix: structural and dynamical effects probed by x-ray absorption spectroscopy.Biophys J. 2007 Feb 15;92(4):1350-60. doi: 10.1529/biophysj.106.092338. Epub 2006 Dec 1. Biophys J. 2007. PMID: 17142287 Free PMC article.
-
The unusually strong hydrogen bond between the carbonyl of Q(A) and His M219 in the Rhodobacter sphaeroides reaction center is not essential for efficient electron transfer from Q(A)(-) to Q(B).Biochemistry. 2007 Jun 5;46(22):6468-76. doi: 10.1021/bi700057f. Epub 2007 May 12. Biochemistry. 2007. PMID: 17497939
-
Coupling of electron transfer to proton uptake at the Q(B) site of the bacterial reaction center: a perspective from FTIR difference spectroscopy.Biochim Biophys Acta. 2008 Oct;1777(10):1229-48. doi: 10.1016/j.bbabio.2008.06.012. Epub 2008 Jul 11. Biochim Biophys Acta. 2008. PMID: 18671937 Review.
-
Internal dynamics and protein-matrix coupling in trehalose-coated proteins.Biochim Biophys Acta. 2005 Jun 1;1749(2):252-81. doi: 10.1016/j.bbapap.2005.03.004. Epub 2005 Apr 15. Biochim Biophys Acta. 2005. PMID: 15886079 Review.
Cited by
-
Carboxylate shifts steer interquinone electron transfer in photosynthesis.J Biol Chem. 2011 Feb 18;286(7):5368-74. doi: 10.1074/jbc.M110.202879. Epub 2010 Dec 17. J Biol Chem. 2011. PMID: 21169354 Free PMC article.
References
-
- Feher, G., J. P. Allen, M. Y. Okamura, and D. C. Rees. 1989. Structure and function of bacterial photosynthetic reaction centres. Nature. 339:111–116.
-
- Kleinfeld, D., M. Y. Okamura, and G. Feher. 1984. Electron-transfer kinetics in photosynthetic reaction centers cooled to cryogenic temperatures in the charge separated state: evidence for light-induced structural changes. Biochemistry. 23:5780–5786. - PubMed
-
- Stowell, M. H. B., T. M. McPhillips, D. C. Rees, S. M. Soltis, E. Abresch, and G. Feher. 1997. Light-induced structural changes in photosynthetic reaction center: implications for mechanism of electron-proton transfer. Science. 276:812–815. - PubMed
-
-
Xu, Q., L. Baciou, P. Sebban, and M. R. Gunner. 2002. Exploring the energy landscape for
to QB electron transfer in bacterial photosynthetic reaction centers: effect of substrate position and tail length on the conformational gating step. Biochemistry. 41:10021–10025. - PubMed
-
Xu, Q., L. Baciou, P. Sebban, and M. R. Gunner. 2002. Exploring the energy landscape for
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

