Synergy and specificity of two Na+-aromatic amino acid symporters in the model alimentary canal of mosquito larvae
- PMID: 18456887
- PMCID: PMC3397476
- DOI: 10.1242/jeb.017244
Synergy and specificity of two Na+-aromatic amino acid symporters in the model alimentary canal of mosquito larvae
Abstract
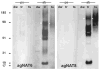
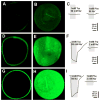
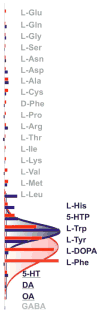
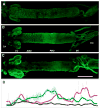
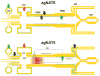
The nutrient amino acid transporter (NAT) subfamily is the largest subdivision of the sodium neurotransmitter symporter family (SNF; also known as SLC6; HUGO). There are seven members of the NAT population in the African malaria mosquito Anopheles gambiae, two of which, AgNAT6 and AgNAT8, preferably transport indole- and phenyl-branched substrates, respectively. The relative expression and distribution of these aromatic NATs were examined with transporter-specific antibodies in Xenopus oocytes and mosquito larval alimentary canal, representing heterologous and tissue expression systems, respectively. NAT-specific aromatic-substrate-induced currents strongly corresponded with specific accumulation of both transporters in the plasma membrane of oocytes. Immunolabeling revealed elevated expressions of both transporters in specific regions of the larval alimentary canal, including salivary glands, cardia, gastric caeca, posterior midgut and Malpighian tubules. Differences in relative expression densities and spatial distribution of the transporters were prominent in virtually all of these regions, suggesting unique profiles of the aromatic amino acid absorption. For the first time reversal of the location of a transporter between apical and basal membranes was identified in posterior and anterior epithelial domains corresponding with secretory and absorptive epithelial functions, respectively. Both aromatic NATs formed putative homodimers in the larval gut whereas functional monomers were over-expressed heterologously in Xenopus oocytes. The results unequivocally suggest functional synergy between substrate-specific AgNAT6 and AgNAT8 in intracellular absorption of aromatic amino acids. More broadly, they suggest that the specific selectivity, regional expression and polarized membrane docking of NATs represent key adaptive traits shaping functional patterns of essential amino acid absorption in the metazoan alimentary canal and other tissues.
Figures

References
-
- Assis P, Boudko DY, Meleshkevitch EA, Phung E. Molecular expression and electrochemical analysis of phenylalanine-tyrosine transporter from Anopheles gambiae larvae. FASEB J. 2004;18:A1269.
-
- Bossi E, Soragna A, Miszner A, Giovannardi S, Frangione V, Peres A. Oligomeric structure of the neutral amino acid transporters KAAT1 and CAATCH1. Am J Physiol. 2007;292:C1379–C1387. - PubMed
-
- Boudko DY, Moroz LL, Linser PJ, Trimarchi JR, Smith PJS, Harvey WR. In situ analysis of pH gradients in mosquito larvae using noninvasive, self-referencing, pH-sensitive microelectrodes. J Exp Biol. 2001b;204:691–699. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

