Characterization of a Helicobacter hepaticus putA mutant strain in host colonization and oxidative stress
- PMID: 18458068
- PMCID: PMC2446744
- DOI: 10.1128/IAI.01737-07
Characterization of a Helicobacter hepaticus putA mutant strain in host colonization and oxidative stress
Abstract
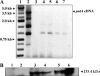
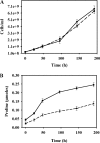
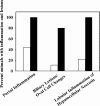
Helicobacter hepaticus is a gram-negative, spiral-shaped microaerophilic bacterium associated with chronic intestinal infection leading to hepatitis and colonic and hepatic carcinomas in susceptible strains of mice. In the closely related human pathogen Helicobacter pylori, L-proline is a preferred respiratory substrate and is found at significantly high levels in the gastric juice of infected patients. A previous study of the proline catabolic PutA flavoenzymes from H. pylori and H. hepaticus revealed that Helicobacter PutA generates reactive oxygen species during proline oxidation by transferring electrons from reduced flavin to molecular oxygen. We further explored the preference for proline as a respiratory substrate and the potential impact of proline metabolism on the redox environment in Helicobacter species during host infection by disrupting the putA gene in H. hepaticus. The resulting putA knockout mutant strain was characterized by oxidative stress analysis and mouse infection studies. The putA mutant strain of H. hepaticus exhibited increased proline levels and resistance to oxidative stress relative to that of the wild-type strain, consistent with proline's role as an antioxidant. The significant increase in stress resistance was attributed to higher proline content, as no upregulation of antioxidant genes was observed for the putA mutant strain. The wild-type and putA mutant H. hepaticus strains displayed similar levels of infection in mice, but in mice challenged with the putA mutant strain, significantly reduced inflammation was observed, suggesting a role for proline metabolism in H. hepaticus pathogenicity in vivo.
Figures
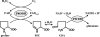


References
-
- Alia, P. Mohanty, and J. Matysik. 2001. Effect of proline on the production of singlet oxygen. Amino Acids 21195-200. - PubMed
-
- Aslan, M., Y. Nazligul, M. Horoz, C. Bolukbas, F. F. Bolukbas, N. Aksoy, H. Celik, and O. Erel. 2007. Serum prolidase activity and oxidative status in Helicobacter pylori infection. Clin. Biochem. 4037-40. - PubMed
-
- Becker, D. F., and E. A. Thomas. 2001. Redox properties of the PutA protein from Escherichia coli and the influence of the flavin redox state on PutA-DNA interactions. Biochemistry 404714-4722. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

