Relative effectiveness of osteoporosis drugs for preventing nonvertebral fracture
- PMID: 18458276
- PMCID: PMC3285566
- DOI: 10.7326/0003-4819-148-9-200805060-00003
Relative effectiveness of osteoporosis drugs for preventing nonvertebral fracture
Abstract
Background: Little information is available on the comparative effectiveness of osteoporosis pharmacotherapies.
Objective: To compare the relative effectiveness of osteoporosis treatments to reduce nonvertebral fracture risk among older adults.
Design: Cohort study.
Setting: Enrollees in 2 statewide pharmaceutical benefit programs for persons age 65 years or older.
Patients: 43,135 new recipients of oral bisphosphonates, nasal calcitonin, and raloxifene who began treatment from 2000 to 2005. The mean age was 79 years (SD, 6.9), and 96% were women.
Measurements: The primary outcome was nonvertebral fracture (hip, humerus, or radius or ulna) within 12 months of treatment initiation. Cox proportional hazard models stratified by state and adjusted for risk factors for fracture were used to compare fracture rates. Alendronate was the reference category in all analyses.
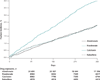
Results: A total of 1051 nonvertebral fractures were observed within 12 months (2.62 fractures per 100 person-years). No large differences in fracture risk were found between risedronate (hazard ratio [HR], 1.01 [95% CI, 0.85 to 1.21]) or raloxifene (HR, 1.18 [CI, 0.96 to 1.46]) and alendronate. However, among those with a fracture history, raloxifene recipients experienced more nonvertebral fractures within 12 months (HR, 1.78 [CI, 1.20 to 2.63]) compared with alendronate recipients. Patients who received calcitonin experienced more nonvertebral fractures than those who received alendronate (HR, 1.40, [CI, 1.20 to 1.63]). Results were similar in sensitivity analyses that examined different lengths of follow-up (6 months and 24 months), were restricted to hip fracture as the outcome, and were completed in various subgroups.
Limitation: Confounder adjustment was limited to health care utilization data, and the confidence bounds of some comparisons were too wide to rule out potential clinically important differences between agents.
Conclusion: Differences in fracture risk between risedronate or raloxifene and alendronate were small. Nasal calcitonin recipients may have a higher risk for nonvertebral fractures compared with alendronate recipients. Future studies that can better adjust for possible confounding may further clarify these relationships.
Conflict of interest statement
Figures



Comment in
-
Comparative efficacy: what we know, what we need to know, and how we can get there.Ann Intern Med. 2008 May 6;148(9):702-3. doi: 10.7326/0003-4819-148-9-200805060-00010. Ann Intern Med. 2008. PMID: 18458283 No abstract available.
-
Did prescription bias affect outcomes in a study of the relative effectiveness of osteoporosis drugs?Ann Intern Med. 2008 Oct 7;149(7):514; author reply 514. doi: 10.7326/0003-4819-149-7-200810070-00017. Ann Intern Med. 2008. PMID: 18838735 No abstract available.
Summary for patients in
-
Summaries for patients. Drug therapy for osteoporosis.Ann Intern Med. 2008 May 6;148(9):I28. doi: 10.7326/0003-4819-148-9-200805060-00002. Ann Intern Med. 2008. PMID: 18458273 No abstract available.
References
-
- Consensus development conference: diagnosis, prophylaxis, and treatment of osteoporosis. Am J Med. 1993;94:646–650. [PMID: 8506892] - PubMed
-
- National Osteoporosis Foundation. Physician’s Guide to Prevention and Treatment of Osteoporosis. Washington, DC: National Osteoporosis Foundation; 2003. [on 30 March 2004]. Accessed at www.nof.org/physguide.html.
-
- Rosen CJ, Hochberg MC, Bonnick SL, McClung M, Miller P, Broy S, et al. Fosamax Actonel Comparison Trial Investigators. Treatment with once-weekly alendronate 70 mg compared with once-weekly risedronate 35 mg in women with postmenopausal osteoporosis: a randomized double-blind study. J Bone Miner Res. 2005;20:141–151. [PMID: 15619680] - PubMed
-
- Bonnick S, Saag KG, Kiel DP, McClung M, Hochberg M, Burnett SM, et al. Comparison of weekly treatment of postmenopausal osteoporosis with alendronate versus risedronate over two years. J Clin Endocrinol Metab. 2006;91:2631–2637. [PMID: 16636120] - PubMed
-
- Luckey M, Kagan R, Greenspan S, Bone H, Kiel RD, Simon J, et al. Once-weekly alendronate 70 mg and raloxifene 60 mg daily in the treatment of postmenopausal osteoporosis. Menopause. 2004;11:405–415. [PMID: 15243278] - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
