Human breast cancer-associated fibroblasts (CAFs) show caveolin-1 downregulation and RB tumor suppressor functional inactivation: Implications for the response to hormonal therapy
- PMID: 18458534
- PMCID: PMC6688494
- DOI: 10.4161/cbt.7.8.6220
Human breast cancer-associated fibroblasts (CAFs) show caveolin-1 downregulation and RB tumor suppressor functional inactivation: Implications for the response to hormonal therapy
Abstract
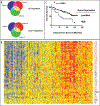
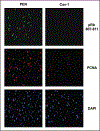
It is becoming increasingly apparent that the tumor microenvironment plays a critical role in human breast cancer onset and progression. Therefore, we isolated cancer-associated fibroblasts (CAFs) from human breast cancer lesions and studied their properties, as compared with normal mammary fibroblasts (NFs) isolated from the same patient. Here, we demonstrate that 8 out of 11 CAFs show dramatic downregulation of caveolin-1 (Cav-1) protein expression; Cav-1 is a well-established marker that is normally decreased during the oncogenic transformation of fibroblasts. Next, we performed gene expression profiling studies (DNA microarray) and established a CAF gene expression signature. Interestingly, the expression signature associated with CAFs encompasses a large number of genes that are regulated via the RB-pathway. The CAF gene signature is also predictive of poor clinical outcome in breast cancer patients that were treated with tamoxifen mono-therapy, indicating that CAFs may be useful for predicting the response to hormonal therapy. Finally, we show that replacement of Cav-1 expression in CAFs (using a cell-permeable peptide approach) is sufficient to revert their hyper-proliferative phenotype and prevent RB hyper-phosphorylation. Taken together, these studies highlight the critical role of Cav-1 downregulation in maintaining the abnormal phenotype of human breast cancer-associated fibroblasts.
Figures





References
-
- Sappino AP, Skalli O, Jackson B, Schurch W, Gabbiani G. Smooth-muscle differentiation in stromal cells of malignant and non-malignant breast tissues. Int J Cancer 1988; 41:707–12. - PubMed
-
- Ronnov-Jessen L, Petersen OW, Bissell MJ. Cellular changes involved in conversion of normal to malignant breast: importance of the stromal reaction. Physiol Rev 1996; 76:69–125. - PubMed
-
- Tlsty TD, Hein PW. Know thy neighbor: stromal cells can contribute oncogenic signals. Curr Opin Genet Dev 2001; 11:54–9. - PubMed
-
- Mueller MM, Fusenig NE. Friends or foes—bipolar effects of the tumour stroma in cancer. Nat Rev Cancer 2004; 4:839–49. - PubMed
-
- Serini G, Gabbiani G. Mechanisms of myofibroblast activity and phenotypic modulation. Exp Cell Res 1999; 250:273–83. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
