The genome sequence of the model ascomycete fungus Podospora anserina
- PMID: 18460219
- PMCID: PMC2441463
- DOI: 10.1186/gb-2008-9-5-r77
The genome sequence of the model ascomycete fungus Podospora anserina
Abstract
Background: The dung-inhabiting ascomycete fungus Podospora anserina is a model used to study various aspects of eukaryotic and fungal biology, such as ageing, prions and sexual development.
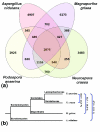
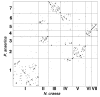
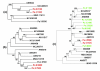
Results: We present a 10X draft sequence of P. anserina genome, linked to the sequences of a large expressed sequence tag collection. Similar to higher eukaryotes, the P. anserina transcription/splicing machinery generates numerous non-conventional transcripts. Comparison of the P. anserina genome and orthologous gene set with the one of its close relatives, Neurospora crassa, shows that synteny is poorly conserved, the main result of evolution being gene shuffling in the same chromosome. The P. anserina genome contains fewer repeated sequences and has evolved new genes by duplication since its separation from N. crassa, despite the presence of the repeat induced point mutation mechanism that mutates duplicated sequences. We also provide evidence that frequent gene loss took place in the lineages leading to P. anserina and N. crassa. P. anserina contains a large and highly specialized set of genes involved in utilization of natural carbon sources commonly found in its natural biotope. It includes genes potentially involved in lignin degradation and efficient cellulose breakdown.
Conclusion: The features of the P. anserina genome indicate a highly dynamic evolution since the divergence of P. anserina and N. crassa, leading to the ability of the former to use specific complex carbon sources that match its needs in its natural biotope.
Figures




References
-
- Hawskworth DL. The magnitude of fungal diversity: the 1.5 million species revisited. Mycol Res. 2001;105:1422–1432.
-
- Bills GF, Christensen M, Powell M, Thorn G. Saprobic soil fungi. In: Mueller GM, Bills GF, Foster MS, editor. Biodiversity of the Fungi, Biodiversity and Monitoring Methods. Amsterdam: Elsevier; 2004. pp. 271–302.
-
- Durrieu G. Ecologie des Champignons. Paris: Masson; 1993.
-
- Money MP. The Triumph of Fungi: a Rotten History. Oxford: Oxford University press; 2007.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

