Functional characterization of HFR1, a high-mannose N-glycan-specific wheat lectin induced by Hessian fly larvae
- PMID: 18467454
- PMCID: PMC2442546
- DOI: 10.1104/pp.108.116145
Functional characterization of HFR1, a high-mannose N-glycan-specific wheat lectin induced by Hessian fly larvae
Abstract
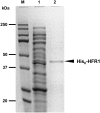
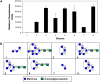
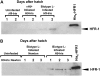
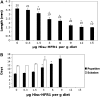
We previously cloned and characterized a novel jacalin-like lectin gene from wheat (Triticum aestivum) plants that responds to infestation by Hessian fly (Mayetiola destructor) larvae, a major dipteran pest of this crop. The infested resistant plants accumulated higher levels of Hfr-1 (for Hessian fly-responsive gene 1) transcripts compared with uninfested or susceptible plants. Here, we characterize the soluble and active recombinant His(6)-HFR1 protein isolated from Escherichia coli. Functional characterization of the protein using hemagglutination assays revealed lectin activity. Glycan microarray-binding assays indicated strong affinity of His(6)-HFR1 to Manalpha1-6(Manalpha1-3)Man trisaccharide structures. Resistant wheat plants accumulated high levels of HFR1 at the larval feeding sites, as revealed by immunodetection, but the avirulent larvae were deterred from feeding and consumed only small amounts of the lectin. Behavioral studies revealed that avirulent Hessian fly larvae on resistant plants exhibited prolonged searching and writhing behaviors as they unsuccessfully attempted to establish feeding sites. During His(6)-HFR1 feeding bioassays, Drosophila melanogaster larvae experienced significant delays in growth and pupation, while percentage mortality increased with progressively higher concentrations of His(6)-HFR1 in the diet. Thus, HFR1 is an antinutrient to dipteran larvae and may play a significant role in deterring Hessian fly larvae from feeding on resistant wheat plants.
Figures



Similar articles
-
Effects of antinutrient proteins on Hessian fly (Diptera: Cecidomyiidae) larvae.J Insect Physiol. 2012 Jan;58(1):41-8. doi: 10.1016/j.jinsphys.2011.09.012. Epub 2011 Sep 29. J Insect Physiol. 2012. PMID: 21983260
-
Hessian fly larval feeding triggers enhanced polyamine levels in susceptible but not resistant wheat.BMC Plant Biol. 2015 Jan 16;15:3. doi: 10.1186/s12870-014-0396-y. BMC Plant Biol. 2015. PMID: 25592131 Free PMC article.
-
A lectin-like wheat gene responds systemically to attempted feeding by avirulent first-instar Hessian fly larvae.J Chem Ecol. 2002 Jul;28(7):1411-28. doi: 10.1023/a:1016200619766. J Chem Ecol. 2002. PMID: 12199504
-
Ultrastructural changes in the midguts of Hessian fly larvae feeding on resistant wheat.J Insect Physiol. 2010 Jul;56(7):754-60. doi: 10.1016/j.jinsphys.2010.01.005. Epub 2010 Feb 4. J Insect Physiol. 2010. PMID: 20116382
-
Gall midges (Hessian flies) as plant pathogens.Annu Rev Phytopathol. 2012;50:339-57. doi: 10.1146/annurev-phyto-072910-095255. Epub 2012 May 29. Annu Rev Phytopathol. 2012. PMID: 22656645 Review.
Cited by
-
Rapid mobilization of membrane lipids in wheat leaf sheaths during incompatible interactions with Hessian fly.Mol Plant Microbe Interact. 2012 Jul;25(7):920-30. doi: 10.1094/MPMI-01-12-0022-R. Mol Plant Microbe Interact. 2012. PMID: 22668001 Free PMC article.
-
Development of SNP assays for hessian fly response genes, Hfr-1 and Hfr-2, for marker-assisted selection in wheat breeding.BMC Genet. 2018 Jul 31;19(1):50. doi: 10.1186/s12863-018-0659-y. BMC Genet. 2018. PMID: 30064355 Free PMC article.
-
Virulent Hessian fly larvae manipulate the free amino acid content of host wheat plants.J Chem Ecol. 2008 Nov;34(11):1401-10. doi: 10.1007/s10886-008-9544-x. Epub 2008 Oct 8. J Chem Ecol. 2008. PMID: 18841417
-
Genetic and phenotypic responses of temperature-independent Hessian fly-resistant durum wheat to larval attack during heat stress.BMC Plant Biol. 2025 Feb 17;25(1):210. doi: 10.1186/s12870-025-06226-1. BMC Plant Biol. 2025. PMID: 39962422 Free PMC article.
-
Ectopic and transient expression of VvDIR4 gene in Arabidopsis and grapes enhances resistance to anthracnose via affecting hormone signaling pathways and lignin production.BMC Genomics. 2024 Sep 28;25(1):895. doi: 10.1186/s12864-024-10830-0. BMC Genomics. 2024. PMID: 39342082 Free PMC article.
References
-
- Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72 248–254 - PubMed
-
- Bunn-Moreno M, Campos-Neto A (1981) Lectin(s) extracted from seeds of Artocarpus integrifolia (jackfruit): potent and selective stimulator(s) of distinct human T and B cell functions. J Immunol 127 427–429 - PubMed
-
- Caliskan M, Cuming AC (2000) Temporal and spatial determination of germin biosynthesis in wheat tissues. Turk J Biol 24 775–782
-
- Chen Y, Peumans WJ, Hause B, Bras J, Kumar M, Proost P, Barre A, Rougé P, Van Damme EJM (2002) Jasmonic acid methyl ester induces the synthesis of a cytoplasmic/nuclear chitooligosaccharide-binding lectin in tobacco leaves. FASEB J 16 905–907 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

