Arrhythmogenic effects of beta2-adrenergic stimulation in the failing heart are attributable to enhanced sarcoplasmic reticulum Ca load
- PMID: 18467626
- PMCID: PMC2585979
- DOI: 10.1161/CIRCRESAHA.107.169011
Arrhythmogenic effects of beta2-adrenergic stimulation in the failing heart are attributable to enhanced sarcoplasmic reticulum Ca load
Abstract
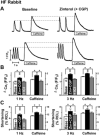
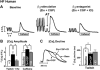
Ventricular tachycardia in heart failure (HF) can initiate by nonreentrant mechanisms such as delayed afterdepolarizations. In an arrhythmogenic rabbit model of HF, we have shown that isoproterenol induces ventricular tachycardia in vivo and aftercontractions and transient inward currents in HF myocytes. To determine whether beta(2)-adrenergic receptor (beta(2)-AR) stimulation contributes, we performed in vivo drug infusion, in vitro myocyte and biochemical studies. Intravenous zinterol (2.5 microg/kg) led to ventricular arrhythmias, including ventricular tachycardia up to 13 beats long in 4 of 6 HF rabbits (versus 0 of 5 controls, P<0.01), an effect blocked by beta(2)-AR antagonist ICI-118,551 (0.2 mg/kg). In field-stimulated myocytes (0.5 to 4 Hz, 37 degrees C), beta(2)-AR stimulation (1 micromol/L zinterol+300 nmol/L beta(1)-AR antagonist CGP-29712A) induced aftercontractions and Ca aftertransients in 88% of HF versus 0% of control myocytes (P<0.01). beta(2)-AR stimulation in HF (but not control) myocytes increased Ca transient amplitude (by 29%), sarcoplasmic reticulum (SR) Ca load (by 28%), the rate of [Ca](i) decline (by 28%; n=12, all P<0.05), and phospholamban phosphorylation at Ser16, but Ca current was unchanged. All of these effects in HF myocytes were blocked by ICI-118,551 (100 nmol/L). Although total beta-AR expression was reduced by 47% in HF rabbit left ventricle, beta(2)-AR number was unchanged, indicating more potent beta(2)-AR-dependent SR Ca uptake and arrhythmogenesis in HF. Human HF myocytes showed similar beta(2)-AR-induced aftercontractions, aftertransients, and enhanced Ca transient amplitude, SR Ca load and twitch [Ca](i) decline rate. Thus, beta(2)-AR stimulation is arrhythmogenic in HF, mediated by SR Ca overload-induced spontaneous SR Ca release and aftercontractions.
Figures






References
-
- Packer M. Sudden unexpected death in patients with congestive heart failure: a second frontier. Circulation. 1985;72(4):681–685. - PubMed
-
- Kjekshus J. Arrhythmias and mortality in congestive heart failure. Am J Cardiol. 1990;65:421–481. - PubMed
-
- Pogwizd SM. Nonreentrant mechanisms underlying spontaneous ventricular arrhythmias in a model of nonischemic heart failure in rabbits. Circulation. 1995;92:1034–1048. - PubMed
-
- Pogwizd SM, McKenzie JP, Cain ME. Mechanisms underlying spontaneous and induced ventricular arrhythmias in patients with idiopathic dilated cardiomyopathy. Circulation. 1998;98:2404–2414. - PubMed
-
- Pogwizd SM, Schlotthaurer K, Li L, Weilong Y, Bers DM. Arrhythmogenesis and contractile dysfunction in heart failure. Circ Res. 2001;88:1159–1167. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

