Decitabine in the treatment of myelodysplastic syndromes
- PMID: 18473005
- PMCID: PMC2376088
Decitabine in the treatment of myelodysplastic syndromes
Abstract
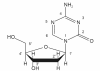
Patients with myelodysplastic syndromes (MDS) are challenging to treat, given the advanced median age and comorbidities of the population. For most patients, the standard therapy is supportive care, including broad-spectrum antibiotics, red blood cell/platelet transfusions, and growth factors. Decitabine, a hypomethylating agent that allows for the re-expression of tumor suppressor genes, represents an exciting new treatment option for MDS patients. In phase 2 and 3 studies, decitabine has been associated with durable responses in MDS patients and delayed time to acute myeloid leukemia (AML) transformation or death compared with supportive care. Decitabine has been shown to be well tolerated with a toxicity profile expected for this class of agent. Recent studies also suggest that lower dose schedules of decitabine may result in additional improvements in response. As more is learned about the mechanism of hypomethylating agents, new roles are emerging for decitabine in combination therapy for MDS and in other hematologic malignancies such as AML.
Keywords: DNA methyltransferase inhibitor; decitabine; gene silencing; hypomethylating agent; myelodysplastic syndromes.
Figures

References
-
- Abele R, Clavel M, Dodion P, et al. The EORTC Early Clinical Trials Cooperative Group experience with 5-aza-2’-deoxycytidine (NSC 127716) in patients with colo-rectal, head and neck, renal carcinomas and malignant melanomas. Eur J Cancer Clin Oncol. 1987;23:1921–4. - PubMed
-
- Alessandrino EP, Amadori S, Barosi G, et al. Evidence- and consensus-based practice guidelines for the therapy of primary myelodysplastic syndromes. A statement from the Italian Society of Hematology. Haematologica. 2002;87:1286–306. - PubMed
-
- American Cancer Society. Multiple myeloma detailed guide. 2006 8/3/06.
-
- American Cancer Society. What are myelodysplastic syndromes and myelodysplastic/myeloproliferative diseases? 2005 19/5/06 L:\\65589.pdf.
-
- Aplastic Anemia & MDS International Foundation. Myelodysplastic syndromes basic explanations. 2005 15/2/06.
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

