Genetic and physiologic dissection of the vertebrate cardiac conduction system
- PMID: 18479184
- PMCID: PMC2430899
- DOI: 10.1371/journal.pbio.0060109
Genetic and physiologic dissection of the vertebrate cardiac conduction system
Abstract
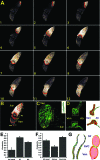
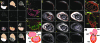
Vertebrate hearts depend on highly specialized cardiomyocytes that form the cardiac conduction system (CCS) to coordinate chamber contraction and drive blood efficiently and unidirectionally throughout the organism. Defects in this specialized wiring system can lead to syncope and sudden cardiac death. Thus, a greater understanding of cardiac conduction development may help to prevent these devastating clinical outcomes. Utilizing a cardiac-specific fluorescent calcium indicator zebrafish transgenic line, Tg(cmlc2:gCaMP)(s878), that allows for in vivo optical mapping analysis in intact animals, we identified and analyzed four distinct stages of cardiac conduction development that correspond to cellular and anatomical changes of the developing heart. Additionally, we observed that epigenetic factors, such as hemodynamic flow and contraction, regulate the fast conduction network of this specialized electrical system. To identify novel regulators of the CCS, we designed and performed a new, physiology-based, forward genetic screen and identified for the first time, to our knowledge, 17 conduction-specific mutations. Positional cloning of hobgoblin(s634) revealed that tcf2, a homeobox transcription factor gene involved in mature onset diabetes of the young and familial glomerulocystic kidney disease, also regulates conduction between the atrium and the ventricle. The combination of the Tg(cmlc2:gCaMP)(s878) line/in vivo optical mapping technique and characterization of cardiac conduction mutants provides a novel multidisciplinary approach to further understand the molecular determinants of the vertebrate CCS.
Conflict of interest statement
Figures







References
-
- Fishman GI. Understanding conduction system development: a hop, skip and jump away. Circ Res. 2005;96:809–811. - PubMed
-
- Pashmforoush M, Lu JT, Chen H, Amand TS, Kondo R, et al. Nkx2–5 pathways and congenital heart disease; loss of ventricular myocyte lineage specification leads to progressive cardiomyopathy and complete heart block. Cell. 2004;117:373–386. - PubMed
-
- Gourdie RG, Mima T, Thompson RP, Mikawa T. Terminal diversification of the myocyte lineage generates Purkinje fibers of the cardiac conduction system. Development. 1995;121:1423–1431. - PubMed
-
- Moskowitz IP, Pizard A, Patel VV, Bruneau BG, Kim JB, et al. The T-box transcription factor Tbx5 is required for the patterning and maturation of the murine cardiac conduction system. Development. 2004;131:4107–4116. - PubMed
-
- Milan DJ, Giokas AC, Serluca FC, Peterson RT, MacRae CA. Notch1b and neuregulin are required for specification of central cardiac conduction tissue. Development. 2006;133:1125–1132. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

