Hypercontractility and impaired sildenafil relaxations in the BKCa channel deletion model of erectile dysfunction
- PMID: 18480246
- PMCID: PMC2494801
- DOI: 10.1152/ajpregu.00173.2008
Hypercontractility and impaired sildenafil relaxations in the BKCa channel deletion model of erectile dysfunction
Abstract
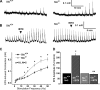
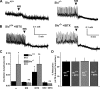
Erectile dysfunction (ED) can be elicited by a variety of pathogenic factors, particularly impaired formation of and responsiveness to nitric oxide (NO) and the downstream effectors soluble guanylate cyclase (sGC) and cGMP-dependent protein kinase I (PKGI). One important target of PKGI in smooth muscle is the large-conductance, Ca2+ -activated potassium (BKCa) channel. In our previous report (42), we demonstrated that deletion of the BKCa channel in mice induced force oscillations and led to reduced nerve-evoked relaxations and ED. In the current study, we used this ED model to explore the role of the BKCa channel in the NO/sGC/PKGI pathway. Electrical field stimulation (EFS)-induced contractions of corpus cavernosum smooth muscle strips were significantly enhanced in the absence of BKCa channel function. In strips precontracted with phenylephrine, EFS-induced relaxations were converted to contractions by inhibition of sGC, and this was further enhanced by loss of BK channel function. Sildenafil-induced relaxations were decreased to a similar extent by inhibition of sGC or BKCa channels. At concentrations >1 microM, sildenafil caused relaxations independent of inhibition of sGC or BKCa channels. Sildenafil did not affect the enhanced force oscillations that were induced by the loss of BKCa channel function. Yet, these oscillations could be completely eliminated by blocking L-type voltage-dependent Ca2+ channels (VDCCs). These results suggest that therapeutically relevant concentrations of sildenafil act through cGMP and BKCa channels, and loss of BKCa channel function leads to hypercontractility, which depends on VDCCs and cannot be modified by the cGMP pathway.
Figures



Similar articles
-
Ca2+ -activated K+ channel (KCa) stimulation improves relaxant capacity of PDE5 inhibitors in human penile arteries and recovers the reduced efficacy of PDE5 inhibition in diabetic erectile dysfunction.Br J Pharmacol. 2013 May;169(2):449-61. doi: 10.1111/bph.12143. Br J Pharmacol. 2013. PMID: 23441682 Free PMC article.
-
Powerful relaxation of phosphodiesterase type 4 inhibitor rolipram in the pig and human bladder neck.J Sex Med. 2014 Apr;11(4):930-941. doi: 10.1111/jsm.12456. Epub 2014 Feb 12. J Sex Med. 2014. PMID: 24754330
-
Inhibitors of phosphodiesterase 5 (PDE 5) inhibit the nerve-induced release of nitric oxide from the rabbit corpus cavernosum.Br J Pharmacol. 2007 Feb;150(3):353-60. doi: 10.1038/sj.bjp.0706991. Epub 2006 Dec 18. Br J Pharmacol. 2007. PMID: 17179943 Free PMC article.
-
[Pharmacological profiles of sildenafil (VIAGRA) in the treatment of erectile dysfunction: efficacy and drug interaction with nitrate].Nihon Yakurigaku Zasshi. 1999 Oct;114(4):213-8. doi: 10.1254/fpj.114.213. Nihon Yakurigaku Zasshi. 1999. PMID: 10584234 Review. Japanese.
-
Phosphodiesterase type 5 as a pharmacologic target in erectile dysfunction.Urology. 2002 Sep;60(2 Suppl 2):4-11. doi: 10.1016/s0090-4295(02)01686-2. Urology. 2002. PMID: 12414329 Review.
Cited by
-
Analysis of erectile responses to H2S donors in the anesthetized rat.Am J Physiol Heart Circ Physiol. 2015 Sep;309(5):H835-43. doi: 10.1152/ajpheart.00293.2015. Epub 2015 Jun 26. Am J Physiol Heart Circ Physiol. 2015. PMID: 26116713 Free PMC article.
-
Molecular and functional characterization of Kv 7 channels in penile arteries and corpus cavernosum of healthy and metabolic syndrome rats.Br J Pharmacol. 2016 May;173(9):1478-90. doi: 10.1111/bph.13444. Epub 2016 Feb 26. Br J Pharmacol. 2016. PMID: 26802314 Free PMC article.
-
Ca2+ -activated K+ channel (KCa) stimulation improves relaxant capacity of PDE5 inhibitors in human penile arteries and recovers the reduced efficacy of PDE5 inhibition in diabetic erectile dysfunction.Br J Pharmacol. 2013 May;169(2):449-61. doi: 10.1111/bph.12143. Br J Pharmacol. 2013. PMID: 23441682 Free PMC article.
-
Emerging gene and stem cell therapies for the treatment of erectile dysfunction.Nat Rev Urol. 2010 Mar;7(3):143-52. doi: 10.1038/nrurol.2010.8. Epub 2010 Feb 16. Nat Rev Urol. 2010. PMID: 20157303 Free PMC article. Review.
-
KCNMA1-linked channelopathy.J Gen Physiol. 2019 Oct 7;151(10):1173-1189. doi: 10.1085/jgp.201912457. Epub 2019 Aug 19. J Gen Physiol. 2019. PMID: 31427379 Free PMC article. Review.
References
-
- Alioua A, Tanaka Y, Wallner M, Hofmann F, Ruth P, Meera P, Toro L. The large conductance, voltage-dependent, and calcium-sensitive K+ channel, Hslo, is a target of cGMP-dependent protein kinase phosphorylation in vivo. J Biol Chem 273: 32950–32956, 1998. - PubMed
-
- Andersson KE, Wagner G. Physiology of penile erection. Physiol Rev 75: 191–236, 1995. - PubMed
-
- Archer SL Potassium channels and erectile dysfunction. Vascul Pharmacol 38: 61–71, 2002. - PubMed
-
- Behr-Roussel D, Gorny D, Mevel K, Compagnie S, Kern P, Sivan V, Bernabe J, Bedigian MP, Alexandre L, Giuliano F. Erectile dysfunction: an early marker for hypertension? A longitudinal study in spontaneously hypertensive rats. Am J Physiol Regul Integr Comp Physiol 288: R276–R283, 2005. - PubMed
-
- Benet AE, Melman A. The epidemiology of erectile dysfunction. Urol Clin North Am 22: 699–709, 1995. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

