Visualization of targeted transduction by engineered lentiviral vectors
- PMID: 18480844
- PMCID: PMC2575058
- DOI: 10.1038/gt.2008.87
Visualization of targeted transduction by engineered lentiviral vectors
Abstract
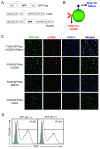
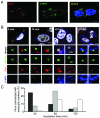
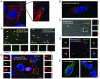
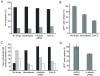
We have reported a method to target lentiviral vectors to specific cell types. This method requires the incorporation of two distinct molecules on the viral vector surface: one is an antibody that renders the targeting specificity for the engineered vector, and the other is a fusogenic protein that allows the engineered vector to enter the target cell. However, the molecular mechanism that controls the targeted infection needs to be defined. In this report, we tracked the individual lentiviral particles by labeling the virus with the GFP-Vpr fusion protein. We were able to visualize the surface-displayed proteins on a single virion as well as antibody-directed targeting to a desired cell type. We also demonstrated the dynamics of virus fusion with endosomes and monitored endosome-associated transport of viruses in target cells. Our results suggest that the fusion between the engineered lentivirus and endosomes takes place at the early endosome level, and that the release of the viral core into the cytosol at the completion of the virus-endosome fusion is correlated with the endosome maturation process. This imaging study sheds some light on the infection mechanism of the engineered lentivirus and can be beneficial to the design of more efficient gene delivery vectors.
Figures

Similar articles
-
Targeting lentiviral vectors to specific cell types in vivo.Proc Natl Acad Sci U S A. 2006 Aug 1;103(31):11479-84. doi: 10.1073/pnas.0604993103. Epub 2006 Jul 24. Proc Natl Acad Sci U S A. 2006. PMID: 16864770 Free PMC article.
-
Retargeting Lentiviruses via SpyCatcher-SpyTag Chemistry for Gene Delivery into Specific Cell Types.mBio. 2017 Dec 12;8(6):e01860-17. doi: 10.1128/mBio.01860-17. mBio. 2017. PMID: 29233896 Free PMC article.
-
Lentiviral Vectors Mediate Long-Term and High Efficiency Transgene Expression in HEK 293T cells.Int J Med Sci. 2015 May 15;12(5):407-15. doi: 10.7150/ijms.11270. eCollection 2015. Int J Med Sci. 2015. PMID: 26005375 Free PMC article.
-
Pseudotyped Lentiviral Vectors: One Vector, Many Guises.Hum Gene Ther Methods. 2017 Dec;28(6):291-301. doi: 10.1089/hgtb.2017.084. Epub 2017 Sep 4. Hum Gene Ther Methods. 2017. PMID: 28870117 Review.
-
Pseudotyping Lentiviral Vectors: When the Clothes Make the Virus.Viruses. 2020 Nov 16;12(11):1311. doi: 10.3390/v12111311. Viruses. 2020. PMID: 33207797 Free PMC article. Review.
Cited by
-
New Advances in Using Virus-like Particles and Related Technologies for Eukaryotic Genome Editing Delivery.Int J Mol Sci. 2022 Aug 6;23(15):8750. doi: 10.3390/ijms23158750. Int J Mol Sci. 2022. PMID: 35955895 Free PMC article. Review.
-
Pseudotyping lentiviral vectors with aura virus envelope glycoproteins for DC-SIGN-mediated transduction of dendritic cells.Hum Gene Ther. 2011 Oct;22(10):1281-91. doi: 10.1089/hum.2010.196. Epub 2011 Jun 13. Hum Gene Ther. 2011. PMID: 21452926 Free PMC article.
-
Virus-receptor mediated transduction of dendritic cells by lentiviruses enveloped with glycoproteins derived from Semliki Forest virus.PLoS One. 2011;6(6):e21491. doi: 10.1371/journal.pone.0021491. Epub 2011 Jun 27. PLoS One. 2011. PMID: 21738680 Free PMC article.
-
Targeted gene delivery to CD117-expressing cells in vivo with lentiviral vectors co-displaying stem cell factor and a fusogenic molecule.Biotechnol Bioeng. 2009 Sep 1;104(1):206-15. doi: 10.1002/bit.22378. Biotechnol Bioeng. 2009. PMID: 19452500 Free PMC article.
-
The Vici syndrome protein EPG5 regulates intracellular nucleic acid trafficking linking autophagy to innate and adaptive immunity.Autophagy. 2018;14(1):22-37. doi: 10.1080/15548627.2017.1389356. Epub 2018 Jan 2. Autophagy. 2018. PMID: 29130391 Free PMC article.
References
-
- Daly G, Chernajovsky Y. Recent developments in retroviral-mediated gene transduction. Mol. Ther. 2000;2:423–434. - PubMed
-
- Mountain A. Gene therapy: the first decade. Trends Biotechnol. 2000;18:119–128. - PubMed
-
- Somia N, Verma IM. Gene therapy: Trials and tribulations. Nat. Rev. Genet. 2000;1:91–99. - PubMed
-
- Verma IM, Somia N. Gene therapy-promises, problems and prospects. Nature. 1997;389:239–242. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

