Estimating glutathione synthesis with deuterated water: a model for peptide biosynthesis
- PMID: 18486587
- PMCID: PMC4065382
- DOI: 10.1016/j.ab.2008.04.042
Estimating glutathione synthesis with deuterated water: a model for peptide biosynthesis
Abstract
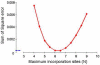
Glutathione (GSH), an intracellular tripeptide that combats oxidative stress, must be continually replaced due to loss through conjugation and destruction. Previous methods, estimating the synthesis of GSH in vivo, used constant infusions of labeled amino acid precursors. We developed a new method based on incorporation of (2)H from orally supplied (2)H(2)O into stable C-H bonds on the tripeptide. The incorporation of (2)H(2)O into GSH was studied in rabbits over a 2-week period. The method estimated N, the maximum number of C-H bonds in GSH that equilibrate with (2)H(2)O as amino acids. GSH was analyzed by liquid chromatography/mass spectrometry after derivatization to yield GSH-N-ethylmaleimide (GSNEM). A model, which simulated the expected abundance at each mass isotopomer for the GSNEM ion at various values for N, was used to find the best fit to the data. The plateau labeling fit best a model with N=6 of a possible 10 C-H bonds. Thus, the amino acid precursors do not completely equilibrate with (2)H(2)O prior to GSH synthesis. Advantages of this new method include replacing costly amino acid infusions with the oral administration of (2)H(2)O and a statistical basis for estimating N.
Figures



References
-
- Han D, Hanawa N, Saberi B, Kaplowitz N. Mechanisms of liver injury. III. Role of glutathione redox status in liver injury. Am.J.Physiol Gastrointest.Liver Physiol. 2006;291:G1–G7. - PubMed
-
- Waelsch H, Rittenberg D. The metabolism of glutathione. Science. 1939;90:423–424. - PubMed
-
- Griffith OW, Meister A. Potent and specific inhibition of glutathione synthesis by buthionine sulfoximine (S-n-butyl homocysteine sulfoximine). J.Biol.Chem. 1979;254:7558–7560. - PubMed
-
- Reid M, Jahoor F. Methods for measuring glutathione concentration and rate of synthesis. Curr.Opin.Clin.Nutr.Metab Care. 2000;3:385–390. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

