Biosynthesis and recycling of nicotinamide cofactors in mycobacterium tuberculosis. An essential role for NAD in nonreplicating bacilli
- PMID: 18490451
- PMCID: PMC2443648
- DOI: 10.1074/jbc.M800694200
Biosynthesis and recycling of nicotinamide cofactors in mycobacterium tuberculosis. An essential role for NAD in nonreplicating bacilli
Abstract
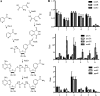
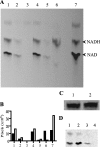
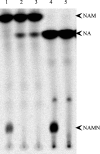
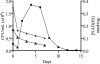
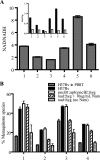
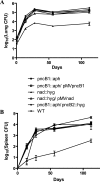
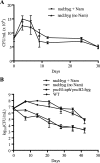
Despite the presence of genes that apparently encode NAD salvage-specific enzymes in its genome, it has been previously thought that Mycobacterium tuberculosis can only synthesize NAD de novo. Transcriptional analysis of the de novo synthesis and putative salvage pathway genes revealed an up-regulation of the salvage pathway genes in vivo and in vitro under conditions of hypoxia. [14C]Nicotinamide incorporation assays in M. tuberculosis isolated directly from the lungs of infected mice or from infected macrophages revealed that incorporation of exogenous nicotinamide was very efficient in in vivo-adapted cells, in contrast to cells grown aerobically in vitro. Two putative nicotinic acid phosphoribosyltransferases, PncB1 (Rv1330c) and PncB2 (Rv0573c), were examined by a combination of in vitro enzymatic activity assays and allelic exchange studies. These studies revealed that both play a role in cofactor salvage. Mutants in the de novo pathway died upon removal of exogenous nicotinamide during active replication in vitro. Cell death is induced by both cofactor starvation and disruption of cellular redox homeostasis as electron transport is impaired by limiting NAD. Inhibitors of NAD synthetase, an essential enzyme common to both recycling and de novo synthesis pathways, displayed the same bactericidal effect as sudden NAD starvation of the de novo pathway mutant in both actively growing and nonreplicating M. tuberculosis. These studies demonstrate the plasticity of the organism in maintaining NAD levels and establish that the two enzymes of the universal pathway are attractive chemotherapeutic targets for active as well as latent tuberculosis.
Figures
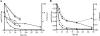
References
-
- Duncan, K., and Barry, C. E., III (2004) Curr. Opin. Microbiol. 7 460–465 - PubMed
-
- Gomez, J. E., and McKinney, J. D. (2004) Tuberculosis (Edinb.) 84 29–44 - PubMed
-
- Boshoff, H. I., and Barry, C. E., III (2005) Nat. Rev. Microbiol. 3 70–80 - PubMed
-
- You, K. S. (1985) CRC Crit. Rev. Biochem. 17 313–451 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information
Molecular Biology Databases

