The peripheral benzodiazepine receptor ligand 1-(2-chlorophenyl-methylpropyl)-3-isoquinoline-carboxamide is a novel antagonist of human constitutive androstane receptor
- PMID: 18492798
- PMCID: PMC2562670
- DOI: 10.1124/mol.108.046656
The peripheral benzodiazepine receptor ligand 1-(2-chlorophenyl-methylpropyl)-3-isoquinoline-carboxamide is a novel antagonist of human constitutive androstane receptor
Abstract
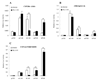
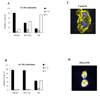
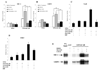
As a promiscuous xenobiotic sensor, the constitutive androstane receptor (CAR; NR1I3) regulates the expression of multiple drug-metabolizing enzymes and transporters in liver. The constitutively activated nature of CAR in the cell-based transfection assays has hindered its use as a predictor of metabolism-based drug-drug interactions. Here, we have identified 1-(2-chlorophenylmethylpropyl)-3-isoquinoline-carboxamide (PK11195), a typical peripheral benzodiazepine receptor (PBR) ligand, as a selective and potent inhibitor of human (h) CAR. In cell-based transfection assays, PK11195 inhibited the constitutive activity of hCAR more than 80% at the concentration of 10 microM, and the PK11195-inhibited activity was efficiently reactivated by the direct CAR activator, 6-(4-chlorophenyl)imidazo[2,1-b][1,3]thiazole-5-carbaldehyde-O-(3,4-dichlorobenzyl) oxime, but not by the indirect hCAR activator, phenobarbital. Mammalian two-hybrid and GST pull-down assays showed that PK11195 repressed the interactions of hCAR with the coactivators steroid receptor coactivator-1 and glucocorticoid receptor-interacting protein 1 to inhibit hCAR activity. The inhibition by PK11195 specifically occurred to the hCAR: PK1195 strongly activated human pregnane X receptor (PXR), whereas it did not alter the activity of the mouse CAR and mouse PXR. In addition, PBR played no role in the PK11195 inhibition of hCAR because the inhibition fully occurred in the HeLa cells in which the PBR was knocked down by small interfering RNA. In the Car(-/-) mouse liver, PK11195 translocated enhanced yellow fluorescent protein-hCAR into the nucleus. These results are consistent with the conclusion that PK11195 is a novel hCAR-specific antagonist that represses the CAR-coactivator interactions to inhibit the receptor activity inside the nucleus. Thus, PK11195 can be used as a chemical tool for studying the molecular basis of CAR function.
Figures





References
-
- Chang C, Norris JD, Gron H, Paige LA, Hamilton PT, Kenan DJ, Fowlkes D, McDonnell DP. Dissection of the LXXLL nuclear receptor-coactivator interaction motif using combinatorial peptide libraries: discovery of peptide antagonists of estrogen receptors alpha and beta. Mol Cell Biol. 1999;19(12):8226–8239. - PMC - PubMed
-
- Faucette SR, Sueyoshi T, Smith CM, Negishi M, Lecluyse EL, Wang H. Differential regulation of hepatic CYP2B6 and CYP3A4 genes by constitutive androstane receptor but not pregnane X receptor. J Pharmacol Exp Ther. 2006;317(3):1200–1209. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

