Heme oxygenase-1 deficiency promotes epithelial-mesenchymal transition and renal fibrosis
- PMID: 18495963
- PMCID: PMC2518436
- DOI: 10.1681/ASN.2007101099
Heme oxygenase-1 deficiency promotes epithelial-mesenchymal transition and renal fibrosis
Abstract
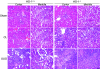
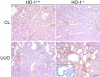
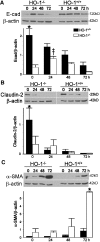
Induction of heme oxygenase-1 (HO-1) is associated with potential antifibrogenic effects. The effects of HO-1 expression on epithelial-mesenchymal transition (EMT), which plays a critical role in the development of renal fibrosis, are unknown. In this study, HO-1(-/-) mice demonstrated significantly more fibrosis after 7 d of unilateral ureteral obstruction compared with wild-type mice, despite similar degrees of hydronephrosis. The obstructed kidneys of HO-1(-/-) mice also had greater macrophage infiltration and renal tubular TGF-beta1 expression than wild-type mice. In addition, the degree of EMT was more extensive in obstructed HO-1(-/-) kidneys, as assessed by alpha-smooth muscle actin and expression of S100A4 in proximal tubular epithelial cells. In vitro studies using proximal tubular cells isolated from HO-1(-/-) and wild-type kidneys confirmed these observations. In conclusion, HO-1 deficiency is associated with increased fibrosis, tubular TGF-beta1 expression, inflammation, and enhanced EMT in obstructive kidney disease. Modulation of the HO-1 pathway may provide a new therapeutic approach to progressive renal diseases.
Figures





Similar articles
-
Intermedin attenuates renal fibrosis by induction of heme oxygenase-1 in rats with unilateral ureteral obstruction.BMC Nephrol. 2017 Jul 11;18(1):232. doi: 10.1186/s12882-017-0659-6. BMC Nephrol. 2017. PMID: 28697727 Free PMC article.
-
Macrophage and epithelial cell H-ferritin expression regulates renal inflammation.Kidney Int. 2015 Jul;88(1):95-108. doi: 10.1038/ki.2015.102. Epub 2015 Apr 15. Kidney Int. 2015. PMID: 25874599 Free PMC article.
-
Membrane rafts-redox signalling pathway contributes to renal fibrosis via modulation of the renal tubular epithelial-mesenchymal transition.J Physiol. 2018 Aug;596(16):3603-3616. doi: 10.1113/JP275952. Epub 2018 Jul 23. J Physiol. 2018. PMID: 29863758 Free PMC article.
-
Induction of heme oxygenase-1 modulates the profibrotic effects of transforming growth factor-beta in human renal tubular epithelial cells.Cell Mol Biol (Noisy-le-grand). 2005 Sep 30;51(4):357-62. Cell Mol Biol (Noisy-le-grand). 2005. PMID: 16309585
-
Adenovirus-mediated P311 ameliorates renal fibrosis through inhibition of epithelial-mesenchymal transition via TGF-β1-Smad-ILK pathway in unilateral ureteral obstruction rats.Int J Mol Med. 2018 May;41(5):3015-3023. doi: 10.3892/ijmm.2018.3485. Epub 2018 Feb 12. Int J Mol Med. 2018. PMID: 29436600
Cited by
-
Heme Oxygenase-1 Induction by Cobalt Protoporphyrin Ameliorates Cholestatic Liver Disease in a Xenobiotic-Induced Murine Model.Int J Mol Sci. 2021 Jul 31;22(15):8253. doi: 10.3390/ijms22158253. Int J Mol Sci. 2021. PMID: 34361019 Free PMC article.
-
Heme oxygenase-1 attenuates epithelial-to-mesenchymal transition of human peritoneal mesothelial cells.Clin Exp Nephrol. 2013 Apr;17(2):284-93. doi: 10.1007/s10157-012-0699-y. Epub 2012 Nov 14. Clin Exp Nephrol. 2013. PMID: 23150184
-
The Triple Crown: NO, CO, and H2S in cancer cell biology.Pharmacol Ther. 2023 Sep;249:108502. doi: 10.1016/j.pharmthera.2023.108502. Epub 2023 Jul 28. Pharmacol Ther. 2023. PMID: 37517510 Free PMC article. Review.
-
Overexpression of Heme Oxygenase-1 Prevents Renal Interstitial Inflammation and Fibrosis Induced by Unilateral Ureter Obstruction.PLoS One. 2016 Jan 14;11(1):e0147084. doi: 10.1371/journal.pone.0147084. eCollection 2016. PLoS One. 2016. PMID: 26765329 Free PMC article.
-
Intermedin attenuates renal fibrosis by induction of heme oxygenase-1 in rats with unilateral ureteral obstruction.BMC Nephrol. 2017 Jul 11;18(1):232. doi: 10.1186/s12882-017-0659-6. BMC Nephrol. 2017. PMID: 28697727 Free PMC article.
References
-
- Klahr S, Morrissey J: Obstructive nephropathy and renal fibrosis. Am J Physiol Renal Physiol 283: F861–F875, 2002 - PubMed
-
- Docherty NG, O’Sullivan OE, Healy DA, Murphy M, O’Neill AJ, Fitzpatrick JM, Watson RW: TGF-β1 induced EMT can occur independently of its pro-apoptotic effects and is aided by EGF receptor activation. Am J Physiol Renal Physiol 290: F1202–F1212, 2006 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

