Positron emission tomography scans obtained for the evaluation of cognitive dysfunction
- PMID: 18514081
- PMCID: PMC4399815
- DOI: 10.1053/j.semnuclmed.2008.02.006
Positron emission tomography scans obtained for the evaluation of cognitive dysfunction
Abstract
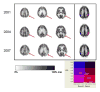
The degree of intactness of human cognitive functioning for a given individual spans a wide spectrum, ranging from normal to severely demented. The differential diagnosis for the causes of impairment along that spectrum is also wide, and often difficult to distinguish clinically, which has led to an increasing role for neuroimaging tools in that evaluation. The most frequent causes of dementia are neurodegenerative disorders, Alzheimer's disease being the most prevalent among them, and they produce significant alterations in brain metabolism, with devastating neuropathologic, clinical, social, and economic consequences. These alterations are detectable through positron emission tomography (PET), even in their earliest stages. The most commonly performed PET studies of the brain are performed with (18)F-fluorodeoxyglucose as the imaged radiopharmaceutical. Such scans have demonstrated diagnostic and prognostic utility for clinicians evaluating patients with cognitive impairment and in distinguishing among primary neurodegenerative disorders and other etiologies contributing to cognitive decline. In addition to focusing on the effects on cerebral metabolism examined with (18)F-fluorodeoxyglucose PET, some other changes occurring in the brains of cognitively impaired patients assessable with other radiotracers will be considered. As preventive and disease-modifying treatments are developed, early detection of accurately diagnosed disease processes facilitated by the use of PET has the potential to substantially impact on the enormous human toll exacted by these diseases.
Figures



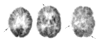
References
-
- Alzheimer's Association. Alzheimer's Disease Facts and Figures. 2007 accessible through www.alz.org.
-
- Powell MR, Smith GE, Knopman DS, et al. Cognitive measures predict pathologic Alzheimer disease. Arch Neurol. 2006;63:865–868. - PubMed
-
- Callahan CM, Hendrie HC, Tierney WM. Documentation and evaluation of cognitive impairment in elderly primary care patients. Ann Intern Med. 1995;122:422–429. - PubMed
-
- Hoffman RS. Diagnostic errors in the evaluation of behavioral disorders. JAMA. 1982;248:964–967. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

