Cardiolipin as an oxidative target in cardiac mitochondria in the aged rat
- PMID: 18515061
- PMCID: PMC2527751
- DOI: 10.1016/j.bbabio.2008.05.444
Cardiolipin as an oxidative target in cardiac mitochondria in the aged rat
Abstract
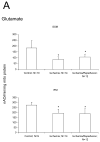
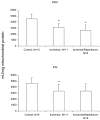
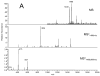
The aged heart sustains greater injury during ischemia (ISC) and reperfusion (REP) compared to the adult heart. In the Fischer 344 (F344) rat, aging decreases oxidative phosphorylation and complex III activity increasing the production of reactive oxygen species in interfibrillar mitochondria (IFM) located among the myofibrils. In the isolated, perfused 24 month old elderly F344 rat heart 25 min of stop-flow ISC causes additional damage to complex III, further decreasing the rate of oxidative phosphorylation. We did not observe further progressive mitochondrial damage during REP. We next asked if ISC or REP increased oxidative damage within mitochondria of the aged heart. Cardiolipin (CL) is a phospholipid unique to mitochondria consisting predominantly of four linoleic acid residues (C18:2). Following ISC and REP in the aged heart, there is a new CL species containing three oxygen atoms added to one linoleic residue. ISC alone was sufficient to generate this new oxidized molecular species of CL. Based upon oxidative damage to CL, complex III activity, and oxidative phosphorylation, mitochondrial damage thus occurs in the aged heart mainly during ISC, rather than during REP. Mitochondrial damage during ischemia sets the stage for mitochondrial-driven cardiomyocyte injury during reperfusion in the aged heart.
Figures



Similar articles
-
Enhanced modification of cardiolipin during ischemia in the aged heart.J Mol Cell Cardiol. 2009 Jun;46(6):1008-15. doi: 10.1016/j.yjmcc.2009.03.007. Epub 2009 Mar 19. J Mol Cell Cardiol. 2009. PMID: 19303420
-
Preservation of cardiolipin content during aging in rat heart interfibrillar mitochondria.J Gerontol A Biol Sci Med Sci. 2002 Jan;57(1):B22-8. doi: 10.1093/gerona/57.1.b22. J Gerontol A Biol Sci Med Sci. 2002. PMID: 11773203
-
Reversal of mitochondrial defects before ischemia protects the aged heart.FASEB J. 2006 Jul;20(9):1543-5. doi: 10.1096/fj.05-4535fje. Epub 2006 Jun 22. FASEB J. 2006. PMID: 16793872
-
Ischemia-reperfusion injury in the aged heart: role of mitochondria.Arch Biochem Biophys. 2003 Dec 15;420(2):287-97. doi: 10.1016/j.abb.2003.09.046. Arch Biochem Biophys. 2003. PMID: 14654068 Review.
-
Mitochondrial Metabolism in Aging Heart.Circ Res. 2016 May 13;118(10):1593-611. doi: 10.1161/CIRCRESAHA.116.307505. Circ Res. 2016. PMID: 27174952 Free PMC article. Review.
Cited by
-
TAZ encodes tafazzin, a transacylase essential for cardiolipin formation and central to the etiology of Barth syndrome.Gene. 2020 Feb 5;726:144148. doi: 10.1016/j.gene.2019.144148. Epub 2019 Oct 21. Gene. 2020. PMID: 31647997 Free PMC article. Review.
-
The role of cardiolipin concentration and acyl chain composition on mitochondrial inner membrane molecular organization and function.Biochim Biophys Acta Mol Cell Biol Lipids. 2019 Jul;1864(7):1039-1052. doi: 10.1016/j.bbalip.2019.03.012. Epub 2019 Apr 2. Biochim Biophys Acta Mol Cell Biol Lipids. 2019. PMID: 30951877 Free PMC article. Review.
-
LC/MS characterization of rotenone induced cardiolipin oxidation in human lymphocytes: implications for mitochondrial dysfunction associated with Parkinson's disease.Mol Nutr Food Res. 2013 Aug;57(8):1410-22. doi: 10.1002/mnfr.201200801. Epub 2013 May 3. Mol Nutr Food Res. 2013. PMID: 23650208 Free PMC article.
-
Age-related decline in mitochondrial bioenergetics: does supercomplex destabilization determine lower oxidative capacity and higher superoxide production?Semin Cell Dev Biol. 2012 Sep;23(7):758-67. doi: 10.1016/j.semcdb.2012.04.002. Epub 2012 Apr 12. Semin Cell Dev Biol. 2012. PMID: 22521482 Free PMC article. Review.
-
Novel therapies targeting inner mitochondrial membrane--from discovery to clinical development.Pharm Res. 2011 Nov;28(11):2669-79. doi: 10.1007/s11095-011-0476-8. Epub 2011 Jun 3. Pharm Res. 2011. PMID: 21638136 Review.
References
-
- Lesnefsky EJ, Lundergan CF, Hodgson JM, Nair R, Reiner JS, Greenhouse SW, Califf RM, Ross AM. Increased left ventricular dysfunction in elderly patients despite successful thrombolysis: the GUSTO-I angiographic experience. J Am Coll Cardiol. 1996;28:331–337. - PubMed
-
- Lesnefsky EJ, Gallo DS, Ye J, Whittingham TS, Lust WD. Aging increases ischemia-reperfusion injury in the isolated, buffer-perfused heart. J Lab Clin Med. 1994;124:843–851. - PubMed
-
- Liu L, Azhar G, Gao W, Zhang X, Wei JY. Bcl-2 and Bax expression in adult rat hearts after coronary occlusion: age-associated differences. Am J Physiol. 1998;275:R315–R322. - PubMed
-
- Tani M, Suganuma Y, Hasegawa H, Shinmura K, Ebihara Y, Hayashi Y, Guo X, Takayama M. Decrease in ischemic tolerance with aging in isolated perfused Fischer 344 rat hearts: relation to increases in intracellular Na+ after ischemia. J Mol Cell Cardiol. 1997;29:3081–3089. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

