Pharmacokinetics of miltefosine in Old World cutaneous leishmaniasis patients
- PMID: 18519729
- PMCID: PMC2493105
- DOI: 10.1128/AAC.00014-08
Pharmacokinetics of miltefosine in Old World cutaneous leishmaniasis patients
Abstract
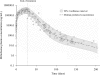
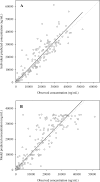
The pharmacokinetics of miltefosine in leishmaniasis patients are, to a great extent, unknown. We examined and characterized the pharmacokinetics of miltefosine in a group of patients with Old World (Leishmania major) cutaneous leishmaniasis. Miltefosine plasma concentrations were determined in samples taken during and up to 5 months after the end of treatment from 31 Dutch military personnel who contracted cutaneous leishmaniasis in Afghanistan and were treated with 150 mg miltefosine/day for 28 days. Samples were analyzed with a validated liquid chromatography-tandem mass spectrometry assay with a lower limit of quantification (LLOQ) of 4 ng/ml. Population pharmacokinetic modeling was performed with nonlinear mixed-effect modeling, using NONMEM. The pharmacokinetics of miltefosine could best be described by an open two-compartment disposition model, with a first elimination half-life of 7.05 days and a terminal elimination half-life of 30.9 days. The median concentration in the last week of treatment (days 22 to 28) was 30,800 ng/ml. The maximum duration of follow-up was 202 days after the start of treatment. All analyzed samples contained a concentration above the LLOQ. Miltefosine is eliminated from the body much slower than previously thought and is therefore still detectable in human plasma samples taken 5 to 6 months after the end of treatment. The presence of subtherapeutic miltefosine concentrations in the blood beyond 5 months after treatment might contribute to the selection of resistant parasites, and moreover, the measures for preventing the teratogenic risks of miltefosine treatment should be reconsidered.
Figures

References
-
- Beal, S. L., and L. B. Sheiner. 1999. NONMEM user's guides. University of California, San Francisco.
-
- Berman, J. 2005. Miltefosine to treat leishmaniasis. Expert Opin. Pharmacother. 6:1381-1388. - PubMed
-
- Berman, J., A. D. M. Bryceson, S. Croft, J. Engel, W. Gutteridge, J. Karbwang, H. Sindermann, J. Soto, S. Sundar, and J. A. Urbina. 2006. Miltefosine: issues to be addressed in the future. Trans. R. Soc. Trop. Med. Hyg. 100(Suppl. 1):S41-S44. - PubMed
-
- Bhattacharya, S. K., T. K. Jha, S. Sundar, C. P. Thakur, J. Engel, H. Sindermann, K. Junge, J. Karbwang, A. D. Bryceson, and J. D. Berman. 2004. Efficacy and tolerability of miltefosine for childhood visceral leishmaniasis in India. Clin. Infect. Dis. 38:217-221. - PubMed
-
- Bhattacharya, S. K., P. K. Sinha, S. Sundar, C. P. Thakur, T. K. Jha, K. Pandey, V. R. Das, N. Kumar, C. Lal, N. Verma, V. P. Singh, A. Ranjan, R. B. Verma, G. Anders, H. Sindermann, and N. K. Ganguly. 2007. Phase 4 trial of miltefosine for the treatment of Indian visceral leishmaniasis. J. Infect. Dis. 196:591-598. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

