Antigen presentation by human microvascular endothelial cells triggers ICAM-1-dependent transendothelial protrusion by, and fractalkine-dependent transendothelial migration of, effector memory CD4+ T cells
- PMID: 18523306
- PMCID: PMC2435282
- DOI: 10.4049/jimmunol.180.12.8386
Antigen presentation by human microvascular endothelial cells triggers ICAM-1-dependent transendothelial protrusion by, and fractalkine-dependent transendothelial migration of, effector memory CD4+ T cells
Abstract
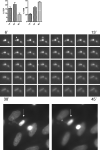
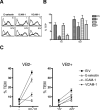
TCR engagement on adherent human effector memory CD4(+) T cells by TNF-treated HUVECs under flow induces formation of a transendothelial protrusion (TEP) by the T cell but fails to induce transendothelial migration (TEM). In contrast, TCR engagement of the same T cell populations by TNF-treated human dermal microvascular cells (HDMEC) not only induces TEP formation, but triggers TEM at or near the interendothelial cell junctions via a process in which TEP formation appears to be the first step. Transduction of adhesion molecules in unactivated HDMEC and use of blocking Abs as conducted with TNF-activated HDMEC indicate that ICAM-1 plays a nonredundant role in TCR-driven TEP formation and TEM, and that TCR-driven TEM is also dependent upon fractalkine. TEP formation, dependence on ICAM-1, and dependence on fractalkine distinguish TCR-induced TEM from IP-10-induced TEM. These in vitro observations suggest that presentation of Ag by human microvascular endothelial cells to circulating CD4(+) effector memory T cells may function to initiate recall responses in peripheral tissues.
Figures




References
-
- Hayry P, von Willebrand E, Andersson LC. Expression of HLA-ABC and -DR locus antigens on human kidney, endothelial, tubular and glomerular cells. Scand J Immunol. 1980;11:303–310. - PubMed
-
- Natali PG, De Martino C, Quaranta V, Nicotra MR, Frezza F, Pellegrino MA, Ferrone S. Expression of Ia-like antigens in normal human nonlymphoid tissues. Transplantation. 1981;31:75–78. - PubMed
-
- Hancock WW, Kraft N, Atkins RC. The immunohistochemical demonstration of major histocompatibility antigens in the human kidney using monoclonal antibodies. Pathology. 1982;14:409–414. - PubMed
-
- Epperson DE, Pober JS. Antigen-presenting function of human endothelial cells. Direct activation of resting CD8 T cells. J Immunol. 1994;153:5402–5412. - PubMed
-
- Dengler TJ, Pober JS. Human vascular endothelial cells stimulate memory but not naive CD8+ T cells to differentiate into CTL retaining an early activation phenotype. J Immunol. 2000;164:5146–5155. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

