A molecular clutch between the actin flow and N-cadherin adhesions drives growth cone migration
- PMID: 18524892
- PMCID: PMC6670336
- DOI: 10.1523/JNEUROSCI.5331-07.2008
A molecular clutch between the actin flow and N-cadherin adhesions drives growth cone migration
Abstract
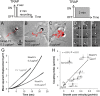
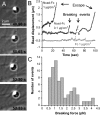
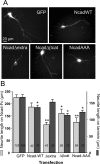
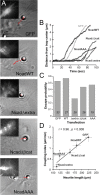
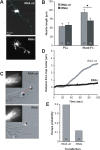
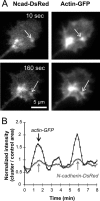
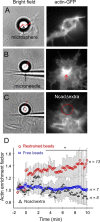
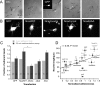
The adhesion molecule N-cadherin plays important roles in the development of the nervous system, in particular by stimulating axon outgrowth, but the molecular mechanisms underlying this effect are mostly unknown. One possibility, the so-called "molecular clutch" model, could involve a direct mechanical linkage between N-cadherin adhesion at the membrane and intracellular actin-based motility within neuronal growth cones. Using live imaging of primary rat hippocampal neurons plated on N-cadherin-coated substrates and optical trapping of N-cadherin-coated microspheres, we demonstrate here a strong correlation between growth cone velocity and the mechanical coupling between ligand-bound N-cadherin receptors and the retrograde actin flow. This relationship holds by varying ligand density and expressing mutated N-cadherin receptors or small interfering RNAs to perturb binding to catenins. By restraining microsphere motion using optical tweezers or a microneedle, we further show slippage of cadherin-cytoskeleton bonds at low forces, and, at higher forces, local actin accumulation, which strengthens nascent N-cadherin contacts. Together, these data support a direct transmission of actin-based traction forces to N-cadherin adhesions, through catenin partners, driving growth cone advance and neurite extension.
Figures

References
-
- Biederer T, Sara Y, Mozhayeva M, Atasoy D, Liu X, Kavalali ET, Sudhof TC. SynCAM, a synaptic adhesion molecule that drives synapse assembly. Science. 2002;297:1525–1531. - PubMed
-
- Boscher C, Mege RM. Cadherin-11 interacts with the FGF receptor and induces neurite outgrowth through associated downstream signalling. Cell Signal. 2008;20:1061–1072. - PubMed
-
- Bozdagi O, Shan W, Tanaka H, Benson DL, Huntley GW. Increasing numbers of synaptic puncta during late-phase LTP: N-cadherin is synthesized, recruited to synaptic sites, and required for potentiation. Neuron. 2000;28:245–259. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
