E-cadherin suppression directs cytoskeletal rearrangement and intraepithelial tumor cell migration in 3D human skin equivalents
- PMID: 18528437
- PMCID: PMC2827766
- DOI: 10.1038/jid.2008.102
E-cadherin suppression directs cytoskeletal rearrangement and intraepithelial tumor cell migration in 3D human skin equivalents
Abstract
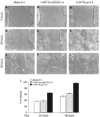
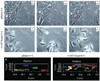
The link between loss of cell-cell adhesion, the activation of cell migration, and the behavior of intraepithelial (IE) tumor cells during the early stages of skin cancer progression is not well understood. The current study characterized the migratory behavior of a squamous cell carcinoma cell line (HaCaT-II-4) upon E-cadherin suppression in both 2D, monolayer cultures and within human skin equivalents that mimic premalignant disease. The migratory behavior of tumor cells was first analyzed in 3D tissue context by developing a model that mimics transepithelial tumor cell migration. We show that loss of cell adhesion enabled migration of single, IE tumor cells between normal keratinocytes as a prerequisite for stromal invasion. To further understand this migratory behavior, E-cadherin-deficient cells were analyzed in 2D, monolayer cultures and displayed altered cytoarchitecture and enhanced membrane protrusive activity that was associated with circumferential actin organization and induction of the nonmuscle, beta actin isoform. These features were associated with increased motility and random, individual cell migration in response to scrape-wounding. Thus, loss of E-cadherin-mediated adhesion led to the acquisition of phenotypic properties that augmented cell motility and directed the transition from the precancer to cancer in skin-like tissues.
Conflict of interest statement
The authors state no conflict of interest.
Figures




References
-
- Allen PG, Shuster CB, Kas J, Chaponnier C, Janmey PA, Herman IM. Phalloidin binding and rheological differences among actin isoforms. Biochemistry. 1996;35:14062–14069. - PubMed
-
- Alt-Holland A, Zhang W, Margulis A, Garlick JA. Microenvironmental control of premalignant disease: the role of intercellular adhesion in the progression of squamous cell carcinoma. Semin Cancer Biol. 2005;15:84–96. - PubMed
-
- Bagutti C, Speight PM, Watt FM. Comparison of integrin, cadherin, and catenin expression in squamous cell carcinomas of the oral cavity. J Pathol. 1998;186:8–16. - PubMed
-
- Behrens J. Cadherins and catenins: role in signal transduction and tumor progression. Cancer Metastasis Rev. 1999;18:15–30. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

