Prospective study of serologic tests for lyme disease
- PMID: 18532885
- PMCID: PMC5538270
- DOI: 10.1086/589242
Prospective study of serologic tests for lyme disease
Abstract
Background: Tests to determine serum antibody levels-the 2-tier sonicate immunoglobulin M (IgM) and immunoglobulin G (IgG) enzyme-linked immunosorbent assay (ELISA) and Western blot method or the IgG of the variable major protein-like sequence-expressed (VlsE) sixth invariant region (C6) peptide ELISA method-are the major tests available for support of the diagnosis of Lyme disease. However, these tests have not been assessed prospectively.
Methods: We used these tests prospectively to determine serologic responses in 134 patients with various manifestations of Lyme disease, 89 patients with other illnesses (with or without a history of Lyme disease), and 136 healthy subjects from areas of endemicity and areas in which the infection was not endemic.
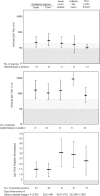
Results: With 2-tier tests and the C6 peptide ELISA, only approximately one-third of 76 patients with erythema migrans had results that were positive for IgM or IgG seroreactivity with Borrelia burgdorferi in acute-phase samples. During convalescence, 3-4 weeks later, almost two-thirds of patients had seroreactivity with the spirochete B. burgdorferi. The frequencies of seroreactivity were significantly greater among patients with spirochetal dissemination than they were among those who lacked evidence of disseminated disease. Of the 44 patients with Lyme disease who had neurologic, heart, or joint involvement, all had positive C6 peptide ELISA results, 42 had IgG responses with 2-tier tests, and 2 patients with facial palsy had only IgM responses. However, among the control groups, the IgG Western blot was slightly more specific than the C6 peptide ELISA. The differences between the 2 test systems (2-tier testing and C6 peptide ELISA) with respect to sensitivity and specificity were not statistically significant.
Conclusions: Except in patients with erythema migrans, both test systems were sensitive for support of the diagnosis of Lyme disease. However, with current methods, 2-tier testing was associated with slightly better specificity.
Conflict of interest statement
Figures
Comment in
-
Editorial commentary: laboratory testing for Lyme disease: time for a change?Clin Infect Dis. 2008 Jul 15;47(2):196-7. doi: 10.1086/589316. Clin Infect Dis. 2008. PMID: 18532894 No abstract available.
-
Serologic tests for lyme disease: more smoke and mirrors.Clin Infect Dis. 2008 Oct 15;47(8):1111-2; author reply 1112-3. doi: 10.1086/592121. Clin Infect Dis. 2008. PMID: 18800935 No abstract available.
References
-
- Steere AC. Lyme disease. N Engl J Med. 2001;345:115–25. - PubMed
-
- Recommendations for test performance and interpretation from the Second International Conference on Serologic Diagnosis of Lyme Disease. MMWR Morb Mortal Wkly Rep. 1995;44:590–1. - PubMed
-
- Case definitions for public health surveillance. MMWR Morb Mortal Wkly Rep. 1990;39(RR-13):1–43. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

