Bone morphogenetic protein regulation of enteric neuronal phenotypic diversity: relationship to timing of cell cycle exit
- PMID: 18537141
- PMCID: PMC2592098
- DOI: 10.1002/cne.21770
Bone morphogenetic protein regulation of enteric neuronal phenotypic diversity: relationship to timing of cell cycle exit
Abstract
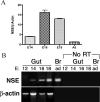
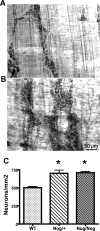
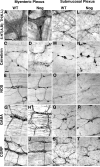
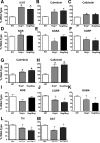
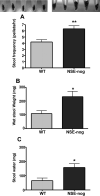
The effects of bone morphogenetic protein (BMP) signaling on enteric neuron development were examined in transgenic mice overexpressing either the BMP inhibitor, noggin, or BMP4 under control of the neuron specific enolase (NSE) promoter. Noggin antagonism of BMP signaling increased total numbers of enteric neurons and those of subpopulations derived from precursors that exit the cell cycle early in neurogenesis (serotonin, calretinin, calbindin). In contrast, noggin overexpression decreased numbers of neurons derived from precursors that exit the cell cycle late (gamma-aminobutyric acid, tyrosine hydroxylase [TH], dopamine transporter, calcitonin gene-related peptide, TrkC). The numbers of TH- and TrkC-expressing neurons were increased by overexpression of BMP4. These observations are consistent with the idea that phenotypic expression in the enteric nervous system (ENS) is determined, in part, by the number of proliferative divisions neuronal precursors undergo before their terminal mitosis. BMP signaling may thus regulate enteric neuronal phenotypic diversity by promoting the exit of precursors from the cell cycle. BMP2 increased the numbers of TH- and TrkC-expressing neurons developing in vitro from immunoselected enteric crest-derived precursors; BMP signaling may thus also specify or promote the development of dopaminergic TrkC/NT-3-dependent neurons. The developmental defects in the ENS of noggin-overexpressing mice caused a relatively mild disturbance of motility (irregular rapid transit and increased stool frequency, weight, and water content). Although the function of the gut thus displays a remarkable tolerance for ENS defects, subtle functional abnormalities in motility or secretion may arise when ENS defects short of aganglionosis occur during development.
(c) 2008 Wiley-Liss, Inc.
Figures



Similar articles
-
Bone morphogenetic protein-2 and -4 limit the number of enteric neurons but promote development of a TrkC-expressing neurotrophin-3-dependent subset.J Neurosci. 2004 Apr 28;24(17):4266-82. doi: 10.1523/JNEUROSCI.3688-03.2004. J Neurosci. 2004. PMID: 15115823 Free PMC article.
-
Neurotrophin-3 in the development of the enteric nervous system.Prog Brain Res. 2004;146:243-63. doi: 10.1016/S0079-6123(03)46016-0. Prog Brain Res. 2004. PMID: 14699968 Review.
-
Pleiotropic effects of the bone morphogenetic proteins on development of the enteric nervous system.Dev Neurobiol. 2012 Jun;72(6):843-56. doi: 10.1002/dneu.22002. Dev Neurobiol. 2012. PMID: 22213745 Free PMC article. Review.
-
Bone morphogenetic proteins regulate enteric gliogenesis by modulating ErbB3 signaling.Dev Biol. 2011 Feb 1;350(1):64-79. doi: 10.1016/j.ydbio.2010.11.017. Epub 2010 Nov 19. Dev Biol. 2011. PMID: 21094638 Free PMC article.
-
Neurotrophin-3 is required for the survival-differentiation of subsets of developing enteric neurons.J Neurosci. 2001 Aug 1;21(15):5620-36. doi: 10.1523/JNEUROSCI.21-15-05620.2001. J Neurosci. 2001. PMID: 11466433 Free PMC article.
Cited by
-
Development and developmental disorders of the enteric nervous system.Nat Rev Gastroenterol Hepatol. 2013 Jan;10(1):43-57. doi: 10.1038/nrgastro.2012.234. Epub 2012 Dec 11. Nat Rev Gastroenterol Hepatol. 2013. PMID: 23229326 Review.
-
The Effect of Microbiota and the Immune System on the Development and Organization of the Enteric Nervous System.Gastroenterology. 2016 Nov;151(5):836-844. doi: 10.1053/j.gastro.2016.07.044. Epub 2016 Aug 10. Gastroenterology. 2016. PMID: 27521479 Free PMC article. Review.
-
Orexinergic modulation of serotonin neurons in the dorsal raphe of a diurnal rodent, Arvicanthis niloticus.Horm Behav. 2019 Nov;116:104584. doi: 10.1016/j.yhbeh.2019.104584. Epub 2019 Aug 31. Horm Behav. 2019. PMID: 31445011 Free PMC article.
-
Cerebral dopamine neurotrophic factor is essential for enteric neuronal development, maintenance, and regulation of gastrointestinal transit.J Comp Neurol. 2020 Oct;528(14):2420-2444. doi: 10.1002/cne.24901. Epub 2020 Mar 26. J Comp Neurol. 2020. PMID: 32154930 Free PMC article.
-
Bone morphogenetic protein 2 regulates the differentiation of nitrergic enteric neurons by modulating Smad1 signaling in slow transit constipation.Mol Med Rep. 2015 Nov;12(5):6547-54. doi: 10.3892/mmr.2015.4297. Epub 2015 Sep 7. Mol Med Rep. 2015. PMID: 26352281 Free PMC article.
References
-
- Anderson RB, Stewart AL, Young HM. Phenotypes of neural-crest-derived cells in vagal and sacral pathways. Cell Tissue Res. 2006;323(1):11–25. - PubMed
-
- Azmitia EC. Modern views on an ancient chemical: serotonin effects on cell proliferation, maturation, and apoptosis. Brain Res Bull. 2001a;56(5):413–424. - PubMed
-
- Azmitia EC. Neuronal instability: implications for Rett's syndrome. Brain Dev. 2001b;23(Suppl 1):S1–S10. - PubMed
-
- Bellier S, Da Silva NR, Aubin-Houzelstein G, Elbaz C, Vanderwinden JM, Panthier JJ. Accelerated intestinal transit in inbred mice with an increased number of interstitial cells of Cajal. Am J Physiol Gastrointest Liver Physiol. 2005;288(1):G151–158. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

