A missense mutation in desmin tail domain linked to human dilated cardiomyopathy promotes cleavage of the head domain and abolishes its Z-disc localization
- PMID: 18539904
- PMCID: PMC2638966
- DOI: 10.1096/fj.07-088724
A missense mutation in desmin tail domain linked to human dilated cardiomyopathy promotes cleavage of the head domain and abolishes its Z-disc localization
Abstract
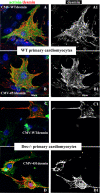
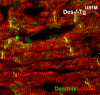
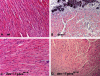
A missense mutation (Ile 451 to Met) at the tail domain of the muscle-specific intermediate filament protein desmin has been suggested to be a genetic cause of dilated cardiomyopathy. The Ile451Met mutation is located inside a conserved motif in the desmin tail domain, believed to have a potential role in the lateral packing of type III intermediate filaments. Nevertheless, the role of the type III intermediate filament tail domain remains elusive. To further study the role of this domain in the function of cardiomyocytes and in the development of cardiomyopathy, we generated transgenic mice expressing the mutant desmin(I451M) in the cardiac tissue. Analysis of hearts from transgenic animals revealed that mutant desmin loses its Z-disc localization but it can still associate with the intercalated discs, which, however, have an altered architecture, resembling other examples of dilated cardiomyopathy. This is the first report demonstrating a critical role of the desmin head and tail domains in the formation of the IF scaffold around Z discs. It is further suggested that in cardiomyocytes, an interplay between desmin tail and head domains is taking place, which potentially protects the amino terminus of desmin from specific proteases. The fact that the association with intercalated discs seems unchanged suggests that this association must take place through the desmin tail, in contrast to the head domain that is most possibly involved in the Z-disc binding.
Figures
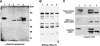





Similar articles
-
Functional characterization of the novel DES mutation p.L136P associated with dilated cardiomyopathy reveals a dominant filament assembly defect.J Mol Cell Cardiol. 2016 Feb;91:207-14. doi: 10.1016/j.yjmcc.2015.12.015. Epub 2015 Dec 23. J Mol Cell Cardiol. 2016. PMID: 26724190
-
Desmin mutation responsible for idiopathic dilated cardiomyopathy.Circulation. 1999 Aug 3;100(5):461-4. doi: 10.1161/01.cir.100.5.461. Circulation. 1999. PMID: 10430757
-
Prevalence of desmin mutations in dilated cardiomyopathy.Circulation. 2007 Mar 13;115(10):1244-51. doi: 10.1161/CIRCULATIONAHA.106.646778. Epub 2007 Feb 26. Circulation. 2007. PMID: 17325244
-
Pathophysiological mechanisms of cardiomyopathies induced by desmin gene variants located in the C-Terminus of segment 2B.J Cell Physiol. 2024 May;239(5):e31254. doi: 10.1002/jcp.31254. Epub 2024 Mar 19. J Cell Physiol. 2024. PMID: 38501553 Review.
-
Desmin filaments and cardiac disease: establishing causality.J Card Fail. 2002 Dec;8(6 Suppl):S287-92. doi: 10.1054/jcaf.2002.129279. J Card Fail. 2002. PMID: 12555134 Review.
Cited by
-
N-acetyl-L-cysteine prevents stress-induced desmin aggregation in cellular models of desminopathy.PLoS One. 2013 Oct 1;8(10):e76361. doi: 10.1371/journal.pone.0076361. eCollection 2013. PLoS One. 2013. PMID: 24098483 Free PMC article.
-
Desminopathies: pathology and mechanisms.Acta Neuropathol. 2013 Jan;125(1):47-75. doi: 10.1007/s00401-012-1057-6. Epub 2012 Nov 11. Acta Neuropathol. 2013. PMID: 23143191 Free PMC article. Review.
-
Crosstalk between coagulation and complement activation promotes cardiac dysfunction in arrhythmogenic right ventricular cardiomyopathy.Theranostics. 2021 Apr 3;11(12):5939-5954. doi: 10.7150/thno.58160. eCollection 2021. Theranostics. 2021. PMID: 33897891 Free PMC article.
-
Increased level of phosphorylated desmin and its degradation products in heart failure.Biochem Biophys Rep. 2016 Feb 27;6:54-62. doi: 10.1016/j.bbrep.2016.02.014. eCollection 2016 Jul. Biochem Biophys Rep. 2016. PMID: 28955862 Free PMC article.
-
Intermediate filaments in cardiomyopathy.Biophys Rev. 2018 Aug;10(4):1007-1031. doi: 10.1007/s12551-018-0443-2. Epub 2018 Jul 19. Biophys Rev. 2018. PMID: 30027462 Free PMC article. Review.
References
-
- Capetanaki Y, Bloch R J, Kouloumenta A, Mavroidis M, Psarras S. Muscle intermediate filaments and their links to membranes and membranous organelles. Exp Cell Res. 2007;313:2063–2076. - PubMed
-
- Li Z, Colucci-Guyon E, Pincon-Raymond M, Mericskay M, Pournin S, Paulin D, Babinet C. Cardiovascular lesions and skeletal myopathy in mice lacking desmin. Dev Biol. 1996;175:362–366. - PubMed
-
- Milner D J, Taffet G E, Wang X, Pham T, Tamura T, Hartley C, Gerdes A M, Capetanaki Y. The absence of desmin leads to cardiomyocyte hypertrophy and cardiac dilation with compromised systolic function. J Mol Cell Cardiol. 1999;31:2063–2076. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

