NK T cells provide lipid antigen-specific cognate help for B cells
- PMID: 18550809
- PMCID: PMC2448838
- DOI: 10.1073/pnas.0801375105
NK T cells provide lipid antigen-specific cognate help for B cells
Abstract
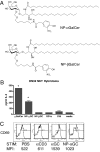
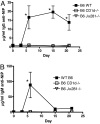
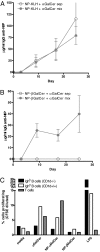
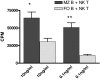
The mechanisms of T cell help for production of antilipid antibodies are largely unknown. This study shows that invariant NK T cells (iNK T cells) and B cells cooperate in a model of antilipid antigen-specific antibody responses. We use a model haptenated lipid molecule, 4-hydroxy-3-nitrophenyl-alphaGalactosylCeramide (NP-alphaGalCer), to demonstrate that iNK T cells provide cognate help to lipid-antigen-presenting B cells. B cells proliferate and IgG anti-NP is produced from in vivo-immunized mice and in vitro cocultures of B and NK T cells after exposure to NP-alphaGalCer, but not closely related control glycolipids. This B cell response is absent in CD1d(-/-) and Jalpha18(-/-) mice but not CD4(-/-) mice. The antibody response to NP-alphaGalCer is dominated by the IgM, IgG3, and IgG2c isotypes, and marginal zone B cells stimulate better in vitro lipid antigen-driven proliferation than follicular B cells, suggesting an important role for this B cell subset. iNK T cell help for B cells is shown to involve cognate help from CD1d-instructed lipid-specific iNK T cells, with help provided via CD40L, B7-1/B7-2, and IFN-gamma, but not IL-4. This model provides evidence of iNK T cell help for antilipid antibody production, an important aspect of infections, autoimmune diseases, and vaccine development. Our findings also now allow prediction of those microbial antigens that would be expected to elicit cognate iNKT cell help for antibody production, namely those that can stimulate iNKT cells and at the same time have a polar moiety that can be recognized by antibodies.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Bendelac A, Bonneville M, Kearney JF. Autoreactivity by design: Innate B and T lymphocytes. Nat Rev Immunol. 2001;1:177–186. - PubMed
-
- Belperron AA, Dailey CM, Bockenstedt LK. Infection-induced marginal zone B cell production of Borrelia hermsii-specific antibody is impaired in the absence of CD1d. J Immunol. 2005;174:5681–5686. - PubMed
-
- Hansen DS, et al. CD1d-restricted NKT cells contribute to malarial splenomegaly and enhance parasite-specific antibody responses. Eur J Immunol. 2003;33:2588–2598. - PubMed
-
- Kobrynski LJ, Sousa AO, Nahmias AJ, Lee FK. Cutting edge: Antibody production to pneumococcal polysaccharides requires CD1 molecules and CD8+ T cells. J Immunol. 2005;174:1787–1790. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

