Basic pharmacodynamic models for agents that alter the lifespan distribution of natural cells
- PMID: 18551354
- PMCID: PMC2673548
- DOI: 10.1007/s10928-008-9092-6
Basic pharmacodynamic models for agents that alter the lifespan distribution of natural cells
Abstract
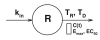
A new class of basic indirect pharmacodynamic models for agents that alter the loss of natural cells based on a lifespan concept are presented. The lifespan indirect response (LIDR) models assume that cells (R) are produced at a constant rate (k(in)), survive during a certain duration T(R), and finally are lost. The rate of cell loss is equal to the production rate but is delayed by T(R). A therapeutic agent can increase or decrease the baseline cell lifespan to a new cell lifespan, T(D), by temporally changing the proportion of cells belonging to the two modes of the lifespan distribution. Therefore, the change of lifespan at time t is described according to the Hill function, H(C(t)), with capacity (E(max)) and sensitivity (EC(50)), and the pharmacokinetic function C(t). A one-compartment cell model was examined through simulations to describe the role of pharmacokinetics, pharmacodynamics and cell properties for the cases where the drug increases (T(D) > T(R)) or decreases (T(D) < T(R)) the cell lifespan. The area under the effect curve (AUCE) and explicit solutions of LIDR models for large doses were derived. The applicability of the model was further illustrated using the effects of recombinant human erythropoietin (rHuEPO) on reticulocytes. The cases of both stimulation of the proliferation of bone marrow progenitor cells and the increase of reticulocyte lifespans were used to describe mean data from healthy subjects who received single subcutaneous doses of rHuEPO ranging from 20 to 160 kIU. rHuEPO is about 4.5-fold less potent in increasing reticulocyte survival than in stimulating the precursor production. A maximum increase of 4.1 days in the mean reticulocyte lifespan was estimated and the effect duration on the lifespan distribution was dose dependent. LIDR models share similar properties with basic indirect response models describing drug stimulation or inhibition of the response loss rate with the exception of the presence of a lag time and a dose independent peak time. The current concept can be applied to describe the pharmacodynamic effects of agents affecting survival of hematopoietic cell populations yielding realistic physiological parameters.
Figures









Similar articles
-
Simultaneous pharmacokinetics/pharmacodynamics modeling of recombinant human erythropoietin upon multiple intravenous dosing in rats.J Pharmacol Exp Ther. 2010 Sep 1;334(3):897-910. doi: 10.1124/jpet.110.167304. Epub 2010 May 25. J Pharmacol Exp Ther. 2010. PMID: 20501635 Free PMC article.
-
Pharmacodynamic models for agents that alter production of natural cells with various distributions of lifespans.J Pharmacokinet Pharmacodyn. 2006 Apr;33(2):125-66. doi: 10.1007/s10928-006-9007-3. Epub 2006 Mar 25. J Pharmacokinet Pharmacodyn. 2006. PMID: 16565883
-
Pharmacodynamic analysis of recombinant human erythropoietin effect on reticulocyte production rate and age distribution in healthy subjects.Clin Pharmacokinet. 2008;47(6):399-415. doi: 10.2165/00003088-200847060-00004. Clin Pharmacokinet. 2008. PMID: 18479174 Free PMC article.
-
Lifespan based indirect response models.J Pharmacokinet Pharmacodyn. 2012 Feb;39(1):109-23. doi: 10.1007/s10928-011-9236-y. Epub 2012 Jan 3. J Pharmacokinet Pharmacodyn. 2012. PMID: 22212685 Free PMC article. Review.
-
Pharmacokinetics/pharmacodynamics of recombinant human erythropoietins in doping control.Sports Med. 2003;33(4):301-15. doi: 10.2165/00007256-200333040-00004. Sports Med. 2003. PMID: 12688828 Review.
Cited by
-
A semi-mechanistic red blood cell survival model provides some insight into red blood cell destruction mechanisms.J Pharmacokinet Pharmacodyn. 2013 Aug;40(4):469-78. doi: 10.1007/s10928-013-9322-4. Epub 2013 Jun 18. J Pharmacokinet Pharmacodyn. 2013. PMID: 23775141
-
Lifespan based pharmacokinetic-pharmacodynamic model of tumor growth inhibition by anticancer therapeutics.PLoS One. 2014 Oct 21;9(10):e109747. doi: 10.1371/journal.pone.0109747. eCollection 2014. PLoS One. 2014. PMID: 25333487 Free PMC article.
-
Simultaneous pharmacokinetics/pharmacodynamics modeling of recombinant human erythropoietin upon multiple intravenous dosing in rats.J Pharmacol Exp Ther. 2010 Sep 1;334(3):897-910. doi: 10.1124/jpet.110.167304. Epub 2010 May 25. J Pharmacol Exp Ther. 2010. PMID: 20501635 Free PMC article.
-
A distributed delay approach for modeling delayed outcomes in pharmacokinetics and pharmacodynamics studies.J Pharmacokinet Pharmacodyn. 2018 Apr;45(2):285-308. doi: 10.1007/s10928-018-9570-4. Epub 2018 Jan 24. J Pharmacokinet Pharmacodyn. 2018. PMID: 29368268
-
Pharmacodynamic modeling of the effect of changes in the environment on cellular lifespan and cellular response.J Pharmacokinet Pharmacodyn. 2008 Oct;35(5):527-52. doi: 10.1007/s10928-008-9100-x. Epub 2008 Oct 21. J Pharmacokinet Pharmacodyn. 2008. PMID: 18937059 Free PMC article.
References
-
- Mager DE, Wyska E, Jusko WJ. Diversity of mechanism-based pharmacodynamic models. Drug Metabol Disp. 2003;31:510–519. - PubMed
-
- Krzyzanski W, Ramakrishnan R, Jusko WJ. Basic pharmacodynamic models for agents that alter production of natural cells. J Pharmacokinet Biopharm. 1993;21:457–478. - PubMed
-
- Ramakrishnan R, Cheung WK, Wacholtz MC, Minton N, Jusko WJ. Pharmacokinetic and pharmacodynamic modeling of recombinant human erythropoietin after single and multiple doses in healthy volunteers. J Clin Pharmacol. 2004;44:991–1002. - PubMed
-
- Krzyzanski W, Jusko WJ, Wacholtz MC, Minton N, Cheung WK. Pharmacokinetic and pharmacodynamic modeling of recombinant human erythropoietin after multiple subcutaneous doses in healthy subjects. Eur J Pharm Sci. 2005;26:295–306. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

