Molecular identity of the late sodium current in adult dog cardiomyocytes identified by Nav1.5 antisense inhibition
- PMID: 18552167
- PMCID: PMC2519192
- DOI: 10.1152/ajpheart.00111.2008
Molecular identity of the late sodium current in adult dog cardiomyocytes identified by Nav1.5 antisense inhibition
Abstract
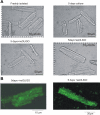
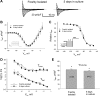
Late Na(+) current (I(NaL)) is a major component of the action potential plateau in human and canine myocardium. Since I(NaL) is increased in heart failure and ischemia, it represents a novel potential target for cardioprotection. However, the molecular identity of I(NaL) remains unclear. We tested the hypothesis that the cardiac Na(+) channel isoform (Na(v)1.5) is a major contributor to I(NaL) in adult dog ventricular cardiomyocytes (VCs). Cultured VCs were exposed to an antisense morpholino-based oligonucleotide (Na(v)1.5 asOligo) targeting the region around the start codon of Na(v)1.5 mRNA or a control nonsense oligonucleotide (nsOligo). Densities of both transient Na(+) current (I(NaT)) and I(NaL) (both in pA/pF) were monitored by whole cell patch clamp. In HEK293 cells expressing Na(v)1.5 or Na(v)1.2, Na(v)1.5 asOligo specifically silenced functional expression of Na(v)1.5 (up to 60% of the initial I(NaT)) but not Na(v)1.2. In both nsOligo-treated controls and untreated VCs, I(NaT) and I(NaL) remained unchanged for up to 5 days. However, both I(NaT) and I(NaL) decreased exponentially with similar time courses (tau = 46 and 56 h, respectively) after VCs were treated with Na(v)1.5 asOligo without changes in 1) decay kinetics, 2) steady-state activation and inactivation, and 3) the ratio of I(NaL) to I(NaT). Four days after exposure to Na(v)1.5 asOligo, I(NaT) and I(NaL) amounted to 68 +/- 6% (mean +/- SE; n = 20, P < 0.01) and 60 +/- 7% (n = 11, P < 0.018) of those in VCs treated by nsOligo, respectively. We conclude that in adult dog heart Na(v)1.5 sodium channels have a "functional half-life" of approximately 35 h (0.69tau) and make a major contribution to I(NaL).
Figures





Similar articles
-
Post-transcriptional silencing of SCN1B and SCN2B genes modulates late sodium current in cardiac myocytes from normal dogs and dogs with chronic heart failure.Am J Physiol Heart Circ Physiol. 2011 Oct;301(4):H1596-605. doi: 10.1152/ajpheart.00948.2009. Epub 2011 Jun 24. Am J Physiol Heart Circ Physiol. 2011. PMID: 21705762 Free PMC article.
-
Late Na+ current produced by human cardiac Na+ channel isoform Nav1.5 is modulated by its beta1 subunit.J Physiol Sci. 2009 May;59(3):217-25. doi: 10.1007/s12576-009-0029-7. Epub 2009 Mar 3. J Physiol Sci. 2009. PMID: 19340536 Free PMC article.
-
Chronic heart failure slows late sodium current in human and canine ventricular myocytes: implications for repolarization variability.Eur J Heart Fail. 2007 Mar;9(3):219-27. doi: 10.1016/j.ejheart.2006.08.007. Epub 2006 Oct 24. Eur J Heart Fail. 2007. PMID: 17067855 Free PMC article.
-
Pathophysiology of the cardiac late Na current and its potential as a drug target.J Mol Cell Cardiol. 2012 Mar;52(3):608-19. doi: 10.1016/j.yjmcc.2011.12.003. Epub 2011 Dec 16. J Mol Cell Cardiol. 2012. PMID: 22198344 Free PMC article. Review.
-
Late sodium current is a new therapeutic target to improve contractility and rhythm in failing heart.Cardiovasc Hematol Agents Med Chem. 2008 Oct;6(4):348-59. doi: 10.2174/187152508785909447. Cardiovasc Hematol Agents Med Chem. 2008. PMID: 18855648 Free PMC article. Review.
Cited by
-
The cardiomyocyte molecular clock, regulation of Scn5a, and arrhythmia susceptibility.Am J Physiol Cell Physiol. 2013 May 15;304(10):C954-65. doi: 10.1152/ajpcell.00383.2012. Epub 2013 Jan 30. Am J Physiol Cell Physiol. 2013. PMID: 23364267 Free PMC article.
-
Post-transcriptional silencing of SCN1B and SCN2B genes modulates late sodium current in cardiac myocytes from normal dogs and dogs with chronic heart failure.Am J Physiol Heart Circ Physiol. 2011 Oct;301(4):H1596-605. doi: 10.1152/ajpheart.00948.2009. Epub 2011 Jun 24. Am J Physiol Heart Circ Physiol. 2011. PMID: 21705762 Free PMC article.
-
Intracellular trafficking pathways of Cx43 gap junction channels.Biochim Biophys Acta Biomembr. 2018 Jan;1860(1):40-47. doi: 10.1016/j.bbamem.2017.05.018. Epub 2017 May 30. Biochim Biophys Acta Biomembr. 2018. PMID: 28576298 Free PMC article. Review.
-
Transmural cellular heterogeneity in myocardial electromechanics.J Physiol Sci. 2018 Jul;68(4):387-413. doi: 10.1007/s12576-017-0541-0. Epub 2017 Jun 1. J Physiol Sci. 2018. PMID: 28573594 Free PMC article.
-
The functional consequences of sodium channel NaV 1.8 in human left ventricular hypertrophy.ESC Heart Fail. 2019 Feb;6(1):154-163. doi: 10.1002/ehf2.12378. Epub 2018 Oct 30. ESC Heart Fail. 2019. PMID: 30378291 Free PMC article.
References
-
- Bridge JH, Smolley J, Spitzer KW, Chin TK. Voltage dependence of sodium-calcium exchange and the control of calcium extrusion in the heart. Ann NY Acad Sci 639: 34–47, 1991. - PubMed
-
- Chattou S, Coulombe A, Diacono J, Le Grand B, John G, Feuvray D. Slowly inactivating component of sodium current in ventricular myocytes is decreased by diabetes and partially inhibited by known Na+-H+ exchange blockers. J Mol Cell Cardiol 32: 1181–1192, 2000. - PubMed
-
- Coraboeuf E, Deroubaix E, Coulombe A. Effect of tetrodotoxin on action potentials of the conducting system in the dog heart. Am J Physiol Heart Circ Physiol 236: H561–H567, 1979. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

