Unique spatial and cellular expression patterns of Hoxa5, Hoxb4, and Hoxb6 proteins in normal developing murine lung are modified in pulmonary hypoplasia
- PMID: 18553509
- PMCID: PMC2670891
- DOI: 10.1002/bdra.20481
Unique spatial and cellular expression patterns of Hoxa5, Hoxb4, and Hoxb6 proteins in normal developing murine lung are modified in pulmonary hypoplasia
Abstract
Background: Hox transcription factors modulate signaling pathways controlling organ morphogenesis and maintain cell fate and differentiation in adults. Retinoid signaling, key in regulating Hox expression, is altered in pulmonary hypoplasia. Information on pattern-specific expression of Hox proteins in normal lung development and in pulmonary hypoplasia is minimal. Our objective was to determine how pulmonary hypoplasia alters temporal, spatial, and cellular expression of Hoxa5, Hoxb4, and Hoxb6 proteins compared to normal lung development.
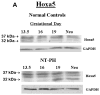
Methods: Temporal, spatial, and cellular Hoxa5, Hoxb4, and Hoxb6 expression was studied in normal (untreated) and nitrofen-induced hypoplastic (NT-PH) lungs from gestational day 13.5, 16, and 19 fetuses and neonates using Western blot and immunohistochemistry.
Results: Modification of protein levels and spatial and cellular Hox expression patterns in NT-PH lungs was consistent with delayed lung development. Distinct protein isoforms were detected for each Hox protein. Expression levels of the Hoxa5 and Hoxb6 protein isoforms changed with development and were altered further in NT-PH lungs. Compared to normal lungs, GD19 and neonatal NT-PH lungs had decreased Hoxb6 and increased Hoxa5 and Hoxb4. Hoxa5 cellular localization changed from mesenchyme to epithelia earlier in normal lungs. Hoxb4 was expressed in mesenchyme and epithelial cells throughout development. Hoxb6 remained mainly in mesenchymal cells around distal airways.
Conclusions: Unique spatial and cellular expression of Hoxa5, Hoxb4, and Hoxb6 participates in branching morphogenesis and terminal sac formation. Altered Hox protein temporal and cellular balance of expression either contributes to pulmonary hypoplasia or functions as a compensatory mechanism attempting to correct abnormal lung development and maturation in this condition.
Copyright 2008 Wiley-Liss, Inc.
Figures












Similar articles
-
Mesenchymal nuclear transcription factors in nitrofen-induced hypoplastic lung.J Surg Res. 2002 Dec;108(2):203-11. doi: 10.1006/jsre.2002.6550. J Surg Res. 2002. PMID: 12505043
-
Expression of T-box transcription factors 2, 4 and 5 is decreased in the branching airway mesenchyme of nitrofen-induced hypoplastic lungs.Pediatr Surg Int. 2017 Feb;33(2):139-143. doi: 10.1007/s00383-016-4005-z. Epub 2016 Nov 11. Pediatr Surg Int. 2017. PMID: 27833996
-
Disturbance of parathyroid hormone-related protein signaling in the nitrofen-induced hypoplastic lung.Pediatr Surg Int. 2010 Jan;26(1):45-50. doi: 10.1007/s00383-009-2506-8. Pediatr Surg Int. 2010. PMID: 19855984
-
Expression of Sproutys and SPREDs is decreased during lung branching morphogenesis in nitrofen-induced pulmonary hypoplasia.Pediatr Surg Int. 2013 Nov;29(11):1193-8. doi: 10.1007/s00383-013-3385-6. Pediatr Surg Int. 2013. PMID: 23982388
-
[Hoxa5: a master gene with multifaceted roles].Med Sci (Paris). 2009 Jan;25(1):77-82. doi: 10.1051/medsci/200925177. Med Sci (Paris). 2009. PMID: 19154698 Review. French.
Cited by
-
Congenital diaphragmatic hernia candidate genes derived from embryonic transcriptomes.Proc Natl Acad Sci U S A. 2012 Feb 21;109(8):2978-83. doi: 10.1073/pnas.1121621109. Epub 2012 Feb 6. Proc Natl Acad Sci U S A. 2012. PMID: 22315423 Free PMC article.
-
Conditional deletion of WT1 in the septum transversum mesenchyme causes congenital diaphragmatic hernia in mice.Elife. 2016 Sep 19;5:e16009. doi: 10.7554/eLife.16009. Elife. 2016. PMID: 27642710 Free PMC article.
-
Regulatory interactions between androgens, Hoxb5, and TGF β signaling in murine lung development.Biomed Res Int. 2013;2013:320249. doi: 10.1155/2013/320249. Epub 2013 Sep 3. Biomed Res Int. 2013. PMID: 24078914 Free PMC article.
-
Clinicopathologic and prognostic implications of HOXA gene and its associated long-noncoding RNAs expression in non-small cell carcinoma: A meta-analysis.Medicine (Baltimore). 2024 Aug 9;103(32):e38087. doi: 10.1097/MD.0000000000038087. Medicine (Baltimore). 2024. PMID: 39121297 Free PMC article.
-
HOXA5 and p53 cooperate to suppress lung cancer cell invasion and serve as good prognostic factors in non-small cell lung cancer.J Cancer. 2017 Apr 9;8(6):1071-1081. doi: 10.7150/jca.17295. eCollection 2017. J Cancer. 2017. PMID: 28529621 Free PMC article.
References
-
- Archavachotikul K, Ciccone TJ, Chinoy MR, Nielsen HC, Volpe MV. Thyroid hormone affects embryonic mouse lung branching morphogenesis and cellular differentiation. Am J Physiol Lung Cell Mol Physiol. 2002;282:L359–L369. - PubMed
-
- Aubin J, De’ry U, Lemieuz M, Chailler P, Jeannotte L. Stomach regional specification requires Hoxa-5 driven mesenchymal-epithelial signaling. Dev. 2002;129:4075–4087. - PubMed
-
- Aubin J, Lemieuz M, Tremblay M, Berard J, Jennotte L. Early postnatal lethality in Hoxa-5 mutant mice is attributable to respiratory tract defects. Dev Biol. 1997;192:432–445. - PubMed
-
- Babiuk RP, Thebaud B, Greer JJ. Reductions in the incidence of nitrofen-induced diaphragmatic hernia by vitamin A and retinoic acid. Am J Physiol Lung Cell Mol Physiol. 2004;286:970–973. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

