Capsule enlargement in Cryptococcus neoformans confers resistance to oxidative stress suggesting a mechanism for intracellular survival
- PMID: 18554313
- PMCID: PMC4405381
- DOI: 10.1111/j.1462-5822.2008.01186.x
Capsule enlargement in Cryptococcus neoformans confers resistance to oxidative stress suggesting a mechanism for intracellular survival
Abstract
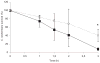
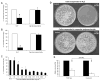
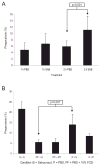
Cryptococcus neoformans is a facultative intracellular pathogen. The most distinctive feature of C. neoformans is a polysaccharide capsule that enlarges depending on environmental stimuli. The mechanism by which C. neoformans avoids killing during phagocytosis is unknown. We hypothesized that capsule growth conferred resistance to microbicidal molecules produced by the host during infection, particularly during phagocytosis. We observed that capsule enlargement conferred resistance to reactive oxygen species produced by H(2)O(2) that was not associated with a higher catalase activity, suggesting a new function for the capsule as a scavenger of reactive oxidative intermediates. Soluble capsular polysaccharide protected C. neoformans and Saccharomyces cerevisiae from killing by H(2)O(2). Acapsular mutants had higher susceptibility to free radicals. Capsular polysaccharide acted as an antioxidant in the nitroblue tetrazolium (NBT) reduction coupled to beta-nicotinamide adenine dinucleotide (NADH)/phenazine methosulfate (PMS) assay. Capsule enlargement conferred resistance to antimicrobial peptides and the antifungal drug Amphotericin B. Interestingly, the capsule had no effect on susceptibility to azoles and increased susceptibility to fluconazole. Capsule enlargement reduced phagocytosis by environmental predators, although we also noticed that in this system, starvation of C. neoformans cells produced resistance to phagocytosis. Our results suggest that capsular enlargement is a mechanism that enhances C. neoformans survival when ingested by phagocytic cells.
Figures




References
-
- Aebi H. Catalase in vitro. Methods Enzymol. 1984;105:121–126. - PubMed
-
- Aksenov SI, Babyeva IP, Golubev VI. On the mechanism of adaptation of micro-organisms to conditions of extreme low humidity. Life Sci Space Res. 1973;11:55–61. - PubMed
-
- Alvarez M, Casadevall A. Phagosome extrusion and host-cell survival after Cryptococcus neoformans phagocytosis by macrophages. Curr Biol. 2006;16:2161–2165. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

