A luciferase immunoprecipitation systems assay enhances the sensitivity and specificity of diagnosis of Strongyloides stercoralis infection
- PMID: 18558872
- PMCID: PMC3379004
- DOI: 10.1086/589718
A luciferase immunoprecipitation systems assay enhances the sensitivity and specificity of diagnosis of Strongyloides stercoralis infection
Abstract
Background: We investigated whether luciferase immunoprecipitation systems (LIPS) can be the basis for a more rapid, specific, and standardized assay for the diagnosis of Strongyloides stercoralis infection.
Methods: A LIPS assay was developed based on immunoglobulin (Ig) G or IgG4 antibody to a recombinant Strongyloides antigen (NIE) and was compared with an NIE enzyme-linked immunosorbent assay (ELISA). A second antigen, S. stercoralis immunoreactive antigen (SsIR), was tested alone and in combination with NIE. The assays were tested using serum samples from patients with parasitologically proven S. stercoralis or filarial infections and from healthy, uninfected control subjects.
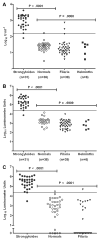
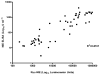
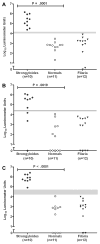
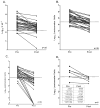
Results: The NIE LIPS assay based on IgG antibody easily differentiated between S. stercoralis-infected and uninfected patients (P< .0001) and demonstrated improved specificity compared with the NIE ELISA (100% vs. 95%). Serum from filaria-infected patients did not cross-react when tested with the NIE LIPS assay. When SsIR was used in combination with NIE in the LIPS format, sensitivity and specificity improved to 100%, with a 7-fold difference between positive and negative values. No advantage was found in using a LIPS assay based on IgG4. At posttreatment follow-up, a significant decline in antibody titers was detected using the NIE ELISA (P< .0017) and the NIE LIPS assay (P< .0001).
Conclusions: LIPS addresses several limitations of current ELISAs and represents a major advance in the diagnosis of S. stercoralis infection.
Conflict of interest statement
Potential conflicts of interest: none reported.
Figures
Similar articles
-
Diagnostic accuracy of a novel enzyme-linked immunoassay for the detection of IgG and IgG4 against Strongyloides stercoralis based on the recombinant antigens NIE/SsIR.Parasit Vectors. 2021 Aug 18;14(1):412. doi: 10.1186/s13071-021-04916-x. Parasit Vectors. 2021. PMID: 34407876 Free PMC article.
-
Improved diagnosis of Strongyloides stercoralis using recombinant antigen-based serologies in a community-wide study in northern Argentina.Clin Vaccine Immunol. 2010 Oct;17(10):1624-30. doi: 10.1128/CVI.00259-10. Epub 2010 Aug 25. Clin Vaccine Immunol. 2010. PMID: 20739501 Free PMC article.
-
Development of Ss-NIE-1 recombinant antigen based assays for immunodiagnosis of strongyloidiasis.PLoS Negl Trop Dis. 2015 Apr 10;9(4):e0003694. doi: 10.1371/journal.pntd.0003694. eCollection 2015 Apr. PLoS Negl Trop Dis. 2015. PMID: 25860665 Free PMC article.
-
Serodiagnosis and early detection of Strongyloides stercoralis infection.J Microbiol Immunol Infect. 2019 Jun;52(3):371-378. doi: 10.1016/j.jmii.2018.10.001. Epub 2018 Oct 11. J Microbiol Immunol Infect. 2019. PMID: 30482708 Review.
-
Strongyloides stercoralis: there but not seen.Curr Opin Infect Dis. 2010 Oct;23(5):500-4. doi: 10.1097/QCO.0b013e32833df718. Curr Opin Infect Dis. 2010. PMID: 20733481 Free PMC article. Review.
Cited by
-
Effectiveness of Strongyloides Recombinant IgG Immunoreactive Antigen in Detecting IgG and IgG4 Subclass Antibodies for Diagnosis of Human Strongyloidiasis Using Rapid Immunochromatographic Tests.Diagnostics (Basel). 2020 Aug 20;10(9):615. doi: 10.3390/diagnostics10090615. Diagnostics (Basel). 2020. PMID: 32825495 Free PMC article.
-
Improved diagnostic sensitivity of human strongyloidiasis using point-of-care mixed recombinant antigen-based immunochromatography.Parasite. 2023;30:60. doi: 10.1051/parasite/2023063. Epub 2023 Dec 13. Parasite. 2023. PMID: 38099622 Free PMC article.
-
Seroprevalence of Strongyloides stercoralis infection in a South Indian adult population.PLoS Negl Trop Dis. 2022 Jul 20;16(7):e0010561. doi: 10.1371/journal.pntd.0010561. eCollection 2022 Jul. PLoS Negl Trop Dis. 2022. PMID: 35857754 Free PMC article.
-
Seroprevalence of the Strongyloides stercoralis Infection in Humans from Yungas Rainforest and Gran Chaco Region from Argentina and Bolivia.Pathogens. 2020 May 20;9(5):394. doi: 10.3390/pathogens9050394. Pathogens. 2020. PMID: 32443925 Free PMC article.
-
IL-10- and TGFβ-mediated Th9 Responses in a Human Helminth Infection.PLoS Negl Trop Dis. 2016 Jan 5;10(1):e0004317. doi: 10.1371/journal.pntd.0004317. eCollection 2016 Jan. PLoS Negl Trop Dis. 2016. PMID: 26730582 Free PMC article.
References
-
- Sato Y, Kobayashi J, Toma H, Shiroma Y. Efficacy of stool examination for detection of Strongyloides infection. Am J Trop Med Hyg. 1995;53:248–50. - PubMed
-
- Conway DJ, Atkins NS, Lillywhite JE, et al. Immunodiagnosis of Strongyloides stercoralis infection: a method for increasing the specificity of the indirect ELISA. Trans R Soc Trop Med Hyg. 1993;87:173– 6. - PubMed
-
- Muck AE, Pires ML, Lammie PJ. Influence of infection with non-filarial helminths on the specificity of serological assays for antifilarial immunoglobulin G4. Trans R Soc Trop Med Hyg. 2003;97:88–90. - PubMed
-
- Ravi V, Ramachandran S, Thompson RW, Andersen JF, Neva FA. Characterization of a recombinant immunodiagnostic antigen (NIE) from Strongyloides stercoralis L3-stage larvae. Mol Biochem Parasitol. 2002;125:73– 81. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

