Shape shifting leads to small-molecule allosteric drug discovery
- PMID: 18559269
- PMCID: PMC2703447
- DOI: 10.1016/j.chembiol.2008.04.012
Shape shifting leads to small-molecule allosteric drug discovery
Abstract
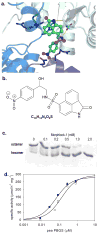
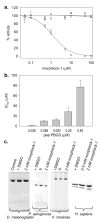
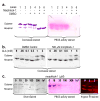
Enzymes that regulate their activity by modulating an equilibrium of alternate, nonadditive, functionally distinct oligomeric assemblies (morpheeins) constitute a recently described mode of allostery. The oligomeric equilibrium for porphobilinogen synthase (PBGS) consists of high-activity octamers, low-activity hexamers, and two dimer conformations. A phylogenetically diverse allosteric site specific to hexamers is proposed as an inhibitor binding site. Inhibitor binding is predicted to draw the oligomeric equilibrium toward the low-activity hexamer. In silico docking enriched a selection from a small-molecule library for compounds predicted to bind to this allosteric site. In vitro testing of selected compounds identified one compound whose inhibition mechanism is species-specific conversion of PBGS octamers to hexamers. We propose that this strategy for inhibitor discovery can be applied to other proteins that use the morpheein model for allosteric regulation.
Figures


Similar articles
-
Porphobilinogen synthase: An equilibrium of different assemblies in human health.Prog Mol Biol Transl Sci. 2020;169:85-104. doi: 10.1016/bs.pmbts.2019.11.003. Epub 2019 Dec 6. Prog Mol Biol Transl Sci. 2020. PMID: 31952692 Free PMC article. Review.
-
Docking to large allosteric binding sites on protein surfaces.Adv Exp Med Biol. 2010;680:481-8. doi: 10.1007/978-1-4419-5913-3_54. Adv Exp Med Biol. 2010. PMID: 20865533 Free PMC article.
-
The Remarkable Character of Porphobilinogen Synthase.Acc Chem Res. 2016 Nov 15;49(11):2509-2517. doi: 10.1021/acs.accounts.6b00414. Epub 2016 Oct 26. Acc Chem Res. 2016. PMID: 27783504 Free PMC article.
-
Allosteric inhibition of human porphobilinogen synthase.J Biol Chem. 2009 Dec 18;284(51):35807-17. doi: 10.1074/jbc.M109.026294. J Biol Chem. 2009. PMID: 19812033 Free PMC article.
-
Allostery and the dynamic oligomerization of porphobilinogen synthase.Arch Biochem Biophys. 2012 Mar 15;519(2):144-53. doi: 10.1016/j.abb.2011.10.010. Epub 2011 Oct 19. Arch Biochem Biophys. 2012. PMID: 22037356 Free PMC article. Review.
Cited by
-
ProtCID: a data resource for structural information on protein interactions.Nat Commun. 2020 Feb 5;11(1):711. doi: 10.1038/s41467-020-14301-4. Nat Commun. 2020. PMID: 32024829 Free PMC article.
-
CytoSIP: an annotated structural atlas for interactions involving cytokines or cytokine receptors.Commun Biol. 2024 May 24;7(1):630. doi: 10.1038/s42003-024-06289-0. Commun Biol. 2024. PMID: 38789577 Free PMC article.
-
Protein acrobatics in pairs--dimerization via domain swapping.Curr Opin Struct Biol. 2009 Feb;19(1):39-49. doi: 10.1016/j.sbi.2008.12.002. Epub 2009 Jan 21. Curr Opin Struct Biol. 2009. PMID: 19162470 Free PMC article. Review. No abstract available.
-
Seesaw protein: Design of a protein that adopts interconvertible alternative functional conformations and its dynamics.Proc Natl Acad Sci U S A. 2025 Feb 18;122(7):e2412117122. doi: 10.1073/pnas.2412117122. Epub 2025 Feb 10. Proc Natl Acad Sci U S A. 2025. PMID: 39928865 Free PMC article.
-
Architecture and assembly of HIV integrase multimers in the absence of DNA substrates.J Biol Chem. 2013 Mar 8;288(10):7373-86. doi: 10.1074/jbc.M112.434431. Epub 2013 Jan 14. J Biol Chem. 2013. PMID: 23322775 Free PMC article.
References
-
- Baeyens KJ, De Bondt HL, Raeymaekers A, Fiers W, De Ranter CJ. The structure of mouse tumour-necrosis factor at 1.4 A resolution: towards modulation of its selectivity and trimerization. Acta Crystallogr D Biol Crystallogr. 1999;55(Pt 4):772–778. - PubMed
-
- Balch WE, Morimoto RI, Dillin A, Kelly JW. Adapting proteostasis for disease intervention. Science. 2008;319(5865):916–919. - PubMed
-
- Battersby AR. Tetrapyrroles: the pigments of life. Nat Prod Rep. 2000;17(6):507–526. - PubMed
-
- Boese QF, Spano AJ, Li JM, Timko MP. Aminolevulinic acid dehydratase in pea (Pisum sativum L.). Identification of an unusual metal-binding domain in the plant enzyme. J Biol Chem. 1991;266(26):17060–17066. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

