The homotetrameric phosphoseryl-tRNA synthetase from Methanosarcina mazei exhibits half-of-the-sites activity
- PMID: 18559342
- PMCID: PMC2494909
- DOI: 10.1074/jbc.M801838200
The homotetrameric phosphoseryl-tRNA synthetase from Methanosarcina mazei exhibits half-of-the-sites activity
Abstract
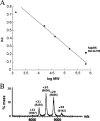
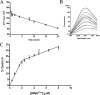
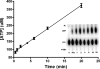
Synthesis of cysteinyl-tRNA(Cys) in methanogenic archaea proceeds by a two-step pathway in which tRNA(Cys) is first aminoacylated with phosphoserine by phosphoseryl-tRNA synthetase (SepRS). Characterization of SepRS from the mesophile Methanosarcina mazei by gel filtration and nondenaturing mass spectrometry shows that the native enzyme exists as an alpha4 tetramer when expressed at high levels in Escherichia coli. However, active site titrations monitored by ATP/PP(i) burst kinetics, together with analysis of tRNA binding stoichiometry by fluorescence spectroscopy, show that the tetrameric enzyme binds two tRNAs and that only two of the four chemically equivalent subunits catalyze formation of phosphoseryl adenylate. Therefore, the phenomenon of half-of-the-sites activity, previously described for synthesis of 1 mol of tyrosyl adenylate by the dimeric class I tyrosyl-tRNA synthetase, operates as well in this homotetrameric class II tRNA synthetase. Analysis of cognate and noncognate reactions by ATP/PP(i) and aminoacylation kinetics strongly suggests that SepRS is able to discriminate against the noncognate amino acids glutamate, serine, and phosphothreonine without the need for a separate hydrolytic editing site. tRNA(Cys) binding to SepRS also enhances the capacity of the enzyme to discriminate among amino acids, indicating the existence of functional connectivity between the tRNA and amino acid binding sites of the enzyme.
Figures

Similar articles
-
Redundant synthesis of cysteinyl-tRNACys in Methanosarcina mazei.J Biol Chem. 2008 Aug 8;283(32):22007-17. doi: 10.1074/jbc.M801839200. Epub 2008 Jun 17. J Biol Chem. 2008. PMID: 18559341 Free PMC article.
-
Toward understanding phosphoseryl-tRNACys formation: the crystal structure of Methanococcus maripaludis phosphoseryl-tRNA synthetase.Proc Natl Acad Sci U S A. 2007 Feb 20;104(8):2620-5. doi: 10.1073/pnas.0611504104. Epub 2007 Feb 14. Proc Natl Acad Sci U S A. 2007. PMID: 17301225 Free PMC article.
-
Pre-transfer editing by class II prolyl-tRNA synthetase: role of aminoacylation active site in "selective release" of noncognate amino acids.J Biol Chem. 2006 Sep 22;281(38):27862-72. doi: 10.1074/jbc.M605856200. Epub 2006 Jul 24. J Biol Chem. 2006. PMID: 16864571
-
Synthetic and editing reactions of aminoacyl-tRNA synthetases using cognate and non-cognate amino acid substrates.Methods. 2017 Jan 15;113:13-26. doi: 10.1016/j.ymeth.2016.09.015. Epub 2016 Oct 3. Methods. 2017. PMID: 27713080 Review.
-
[Superspecificity of aminoacyl-tRNA-synthases].Mol Biol (Mosk). 1984 Jan-Feb;18(1):205-26. Mol Biol (Mosk). 1984. PMID: 6423966 Review. Russian.
Cited by
-
Genetically Encoded Protein Phosphorylation in Mammalian Cells.Cell Chem Biol. 2018 Sep 20;25(9):1067-1074.e5. doi: 10.1016/j.chembiol.2018.05.013. Epub 2018 Jun 21. Cell Chem Biol. 2018. PMID: 29937407 Free PMC article.
-
Redundant synthesis of cysteinyl-tRNACys in Methanosarcina mazei.J Biol Chem. 2008 Aug 8;283(32):22007-17. doi: 10.1074/jbc.M801839200. Epub 2008 Jun 17. J Biol Chem. 2008. PMID: 18559341 Free PMC article.
-
Ancient translation factor is essential for tRNA-dependent cysteine biosynthesis in methanogenic archaea.Proc Natl Acad Sci U S A. 2014 Jul 22;111(29):10520-5. doi: 10.1073/pnas.1411267111. Epub 2014 Jul 7. Proc Natl Acad Sci U S A. 2014. PMID: 25002468 Free PMC article.
-
TrmD: A Methyl Transferase for tRNA Methylation With m1G37.Enzymes. 2017;41:89-115. doi: 10.1016/bs.enz.2017.03.003. Epub 2017 Apr 12. Enzymes. 2017. PMID: 28601227 Free PMC article. Review.
-
Crystallographic analysis of a subcomplex of the transsulfursome with tRNA for Cys-tRNA(Cys) synthesis.Acta Crystallogr F Struct Biol Commun. 2016 Jul;72(Pt 7):569-72. doi: 10.1107/S2053230X16009559. Epub 2016 Jun 28. Acta Crystallogr F Struct Biol Commun. 2016. PMID: 27380375 Free PMC article.
References
-
- Ibba, M., and Söll, D. (2004) Genes Dev. 18 731–738 - PubMed
-
- Sauerwald, A., Zhu, W., Major, T., Roy, H., Palioura, S., Jahn, D., Whitman, W. B., Yates, J. R., III, Ibba, M., and Söll, D. (2005) Science 307 1969–1972 - PubMed
-
- Tumbula, D. L., Becker, H. D., Chang, W. Z., and Söll, D. (2000) Nature 407 106–110 - PubMed
-
- Hanke, T., Bartmann, P., Hennecke, H., Kosakowski, H. M., Jaenicke, R., Holler, E., and Bock, A. (1974) Eur. J. Biochem. 43 601–607 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

