Pharmacokinetics of curcumin conjugate metabolites in healthy human subjects
- PMID: 18559556
- PMCID: PMC4138955
- DOI: 10.1158/1055-9965.EPI-07-2693
Pharmacokinetics of curcumin conjugate metabolites in healthy human subjects
Abstract
Background: Curcumin is a polyphenol, found in the spice turmeric, that has promising anticancer properties, but previous studies suggest that absorption of curcumin may be limited.
Methods: This study examined the pharmacokinetics of a curcumin preparation in healthy human volunteers 0.25 to 72 h after a single oral dose. Curcumin was administered at doses of 10 g (n = 6) and 12 g (n = 6). Subjects were randomly allocated to dose level for a total of six subjects at each dose level. Serum samples were assayed for free curcumin, for its glucuronide, and for its sulfate conjugate. The data were fit to a one-compartment absorption and elimination model.
Results: Using a high-performance liquid chromatography assay with a limit of detection of 50 ng/mL, only one subject had detectable free curcumin at any of the 14 time points assayed, but curcumin glucuronides and sulfates were detected in all subjects. Based on the pharmacokinetic model, the area under the curve for the 10 and 12 g doses was estimated (mean +/- SE) to be 35.33 +/- 3.78 and 26.57 +/- 2.97 mug/mL x h, respectively, whereas C(max) was 2.30 +/- 0.26 and 1.73 +/- 0.19 mug/mL. The T(max) and t(1/2) were estimated to be 3.29 +/- 0.43 and 6.77 +/- 0.83 h. The ratio of glucuronide to sulfate was 1.92:1. The curcumin conjugates were present as either glucuronide or sulfate, not mixed conjugates.
Conclusion: Curcumin is absorbed after oral dosing in humans and can be detected as glucuronide and sulfate conjugates in plasma.
Figures

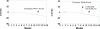

References
-
- Cragg GM, Newman DJ, Snader KM. Natural products in drug discovery and development. J Nat Prod. 1997 Jan;60(1):52–60. - PubMed
-
- Newman DJ, Cragg GM, Holbeck S, Sausville EA. Natural products and derivatives as leads to cell cycle pathway targets in cancer chemotherapy. Curr Cancer Drug Targets. 2002 Dec;2(4):279–308. - PubMed
-
- Newman DJ, Cragg GM, Snader KM. The influence of natural products upon drug discovery. Nat Prod Rep. 2000 Jun;17(3):215–34. - PubMed
-
- Jemal A, Murray T, Ward E, et al. Cancer statistics, 2005. CA: A Cancer Journal for Clinicians. 2005 Jan-Feb;55(1):10–30. - PubMed
-
- Conney AH, Lysz T, Ferraro T, et al. Inhibitory effect of curcumin and some related dietary compounds on tumor promotion and arachidonic acid metabolism in mouse skin. Adv Enzyme Regul. 1991;31:385–96. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

