Tumor trailing strategy for intensity-modulated radiation therapy of moving targets
- PMID: 18561647
- PMCID: PMC2809728
- DOI: 10.1118/1.2900108
Tumor trailing strategy for intensity-modulated radiation therapy of moving targets
Abstract
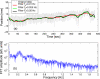
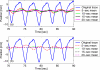
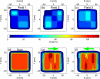
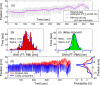
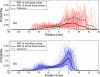
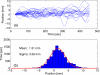
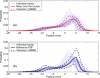
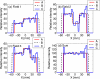
Internal organ motion during the course of radiation therapy of cancer affects the distribution of the delivered dose and, generally, reduces its conformality to the targeted volume. Previously proposed approaches aimed at mitigating the effect of internal motion in intensity-modulated radiation therapy (IMRT) included expansion of the target margins, motion-correlated delivery (e.g., respiratory gating, tumor tracking), and adaptive treatment plan optimization employing a probabilistic description of motion. We describe and test the tumor trailing strategy, which utilizes the synergy of motion-adaptive treatment planning and delivery methods. We regard the (rigid) target motion as a superposition of a relatively fast cyclic component (e.g., respiratory) and slow aperiodic trends (e.g., the drift of exhalation baseline). In the trailing approach, these two components of motion are decoupled and dealt with separately. Real-time motion monitoring is employed to identify the "slow" shifts, which are then corrected by applying setup adjustments. The delivery does not track the target position exactly, but trails the systematic trend due to the delay between the time a shift occurs, is reliably detected, and, subsequently, corrected. The "fast" cyclic motion is accounted for with a robust motion-adaptive treatment planning, which allows for variability in motion parameters (e.g., mean and extrema of the tidal volume, variable period of respiration, and expiratory duration). Motion-surrogate data from gated IMRT treatments were used to provide probability distribution data for motion-adaptive planning and to test algorithms that identified systematic trends in the character of motion. Sample IMRT fields were delivered on a clinical linear accelerator to a programmable moving phantom. Dose measurements were performed with a commercial two-dimensional ion-chamber array. The results indicate that by reducing intrafractional motion variability, the trailing strategy enhances relevance and applicability of motion-adaptive planning methods, and improves conformality of the delivered dose to the target in the presence of irregular motion. Trailing strategy can be applied to respiratory-gated treatments, in which the correction for the slow motion can increase the duty cycle, while robust probabilistic planning can improve management of the residual motion within the gate window. Similarly, trailing may improve the dose conformality in treatment of patients who exhibit detectable target motion of low amplitude, which is considered insufficient to provide a clinical indication for the use of respiratory-gated treatment (e.g., peak-to-peak motion of less than 10 mm). The mechanical limitations of implementing tumor trailing are less rigorous than those of real-time tracking, and the same technology could be used for both.
Figures


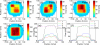


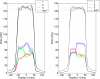
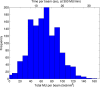

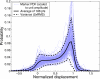
Similar articles
-
Dosimetric evaluation of MRI-guided multi-leaf collimator tracking and trailing for lung stereotactic body radiation therapy.Med Phys. 2021 Apr;48(4):1520-1532. doi: 10.1002/mp.14772. Epub 2021 Mar 12. Med Phys. 2021. PMID: 33583042 Free PMC article.
-
Tumor Trailing for Liver SBRT on the MR-Linac.Int J Radiat Oncol Biol Phys. 2019 Feb 1;103(2):468-478. doi: 10.1016/j.ijrobp.2018.09.011. Epub 2018 Sep 20. Int J Radiat Oncol Biol Phys. 2019. PMID: 30243573
-
Experimental evaluation of a robust optimization method for IMRT of moving targets.Phys Med Biol. 2009 May 7;54(9):2901-14. doi: 10.1088/0031-9155/54/9/021. Epub 2009 Apr 21. Phys Med Biol. 2009. PMID: 19384003
-
Intrafractional motion management in external beam radiotherapy.J Xray Sci Technol. 2019;27(6):1071-1086. doi: 10.3233/XST-180472. J Xray Sci Technol. 2019. PMID: 31476194 Review.
-
Robust radiotherapy planning.Phys Med Biol. 2018 Nov 12;63(22):22TR02. doi: 10.1088/1361-6560/aae659. Phys Med Biol. 2018. PMID: 30418942 Review.
Cited by
-
Evaluation and commissioning of a surface based system for respiratory sensing in 4D CT.J Appl Clin Med Phys. 2010 Dec 4;12(1):3288. doi: 10.1120/jacmp.v12i1.3288. J Appl Clin Med Phys. 2010. PMID: 21330975 Free PMC article.
-
Particle therapy of moving targets-the strategies for tumour motion monitoring and moving targets irradiation.Br J Radiol. 2016 Oct;89(1066):20150275. doi: 10.1259/bjr.20150275. Epub 2016 Jul 19. Br J Radiol. 2016. PMID: 27376637 Free PMC article. Review.
-
Use of dMLC for implementation of dynamic respiratory-gated radiation therapy.Med Phys. 2013 Oct;40(10):101708. doi: 10.1118/1.4820534. Med Phys. 2013. PMID: 24089898 Free PMC article.
-
Potential underestimation of the internal target volume (ITV) from free-breathing CBCT.Med Phys. 2011 Aug;38(8):4689-99. doi: 10.1118/1.3613153. Med Phys. 2011. PMID: 21928643 Free PMC article.
-
4D-MRI driven MR-guided online adaptive radiotherapy for abdominal stereotactic body radiation therapy on a high field MR-Linac: Implementation and initial clinical experience.Clin Transl Radiat Oncol. 2020 May 15;23:72-79. doi: 10.1016/j.ctro.2020.05.002. eCollection 2020 Jul. Clin Transl Radiat Oncol. 2020. PMID: 32490218 Free PMC article.
References
-
- Keall P. J., Kini V. R., Vedam S. S., and Mohan R., “Potential radiotherapy improvements with respiratory gating,” Australas. Phys. Eng. Sci. Med. 25, 1–6 (2002). - PubMed
-
- Bortfeld T., Jiang S. B., and Rietzel E., “Effects of motion on the total dose distribution,” Semin. Radiat. Oncol. 14, 41–51 (2004). - PubMed
-
- Mori S., Chen G. T. Y., and Endo M., “Effects of intrafractional motion on water equivalent pathlength in respiratory-gated heavy charged particle beam radiotherapy,” Int. J. Radiat. Oncol., Biol., Phys. 69, 308–317 (2007). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

