Human respiratory syncytial virus glycoproteins are not required for apical targeting and release from polarized epithelial cells
- PMID: 18562526
- PMCID: PMC2519684
- DOI: 10.1128/JVI.00827-08
Human respiratory syncytial virus glycoproteins are not required for apical targeting and release from polarized epithelial cells
Abstract
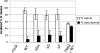
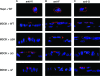
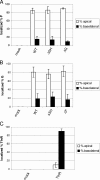
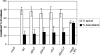
Human respiratory syncytial virus (HRSV) is released from the apical membrane of polarized epithelial cells. However, little is known about the processes of assembly and release of HRSV and which viral gene products are involved in the directional maturation of the virus. Based on previous studies showing that the fusion (F) glycoprotein contained an intrinsic apical sorting signal and that N- and O-linked glycans can act as apical targeting signals, we investigated whether the glycoproteins of HRSV were involved in its directional targeting and release. We generated recombinant viruses with each of the three glycoprotein genes deleted individually or in groups. Each deleted gene was replaced with a reporter gene to maintain wild-type levels of gene expression. The effects of deleting the glycoprotein genes on apical maturation and on targeting of individual proteins in polarized epithelial cells were examined by using biological, biochemical, and microscopic assays. The results of these studies showed that the HRSV glycoproteins are not required for apical maturation or release of the virus. Further, deletion of one or more of the glycoprotein genes did not affect the intracellular targeting of the remaining viral glycoproteins or the nucleocapsid protein to the apical membrane.
Figures



References
-
- Arumugham, R. G., S. W. Hildreth, and P. R. Paradiso. 1989. Interprotein disulfide bonding between F and G glycoproteins of human respiratory syncytial virus. Arch. Virol. 10565-79. - PubMed
-
- Barman, S., L. Adhikary, Y. Kawaoka, and D. P. Nayak. 2003. Influenza A virus hemagglutinin containing basolateral localization signal does not alter the apical budding of a recombinant influenza A virus in polarized MDCK cells. Virology 305138-152. - PubMed
-
- Barr, J. N., S. P. Whelan, and G. W. Wertz. 2002. Transcriptional control of the RNA-dependent RNA polymerase of vesicular stomatitis virus. Biochim. Biophys. Acta 1577337-353. - PubMed
-
- Boulan, E. R., and M. Pendergast. 1980. Polarized distribution of viral envelope proteins in the plasma membrane of infected epithelial cells. Cell 2045-54. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

