A replication-competent, neuronal spread-defective, live attenuated herpes simplex virus type 1 vaccine
- PMID: 18562543
- PMCID: PMC2519657
- DOI: 10.1128/JVI.00551-08
A replication-competent, neuronal spread-defective, live attenuated herpes simplex virus type 1 vaccine
Abstract
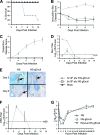
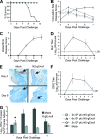
Herpes simplex virus type 1 (HSV-1) produces oral lesions, encephalitis, keratitis, and severe infections in the immunocompromised host. HSV-1 is almost as common as HSV-2 in causing first episodes of genital herpes, a disease that is associated with an increased risk of human immunodeficiency virus acquisition and transmission. No approved vaccines are currently available to protect against HSV-1 or HSV-2 infection. We developed a novel HSV vaccine strategy that uses a replication-competent strain of HSV-1, NS-gEnull, which has a defect in anterograde and retrograde directional spread and cell-to-cell spread. Following scratch inoculation on the mouse flank, NS-gEnull replicated at the site of inoculation without causing disease. Importantly, the vaccine strain was not isolated from dorsal root ganglia (DRG). We used the flank model to challenge vaccinated mice and demonstrated that NS-gEnull was highly protective against wild-type HSV-1. The challenge virus replicated to low titers at the site of inoculation; therefore, the vaccine strain did not provide sterilizing immunity. Nevertheless, challenge by HSV-1 or HSV-2 resulted in less-severe disease at the inoculation site, and vaccinated mice were totally protected against zosteriform disease and death. After HSV-1 challenge, latent virus was recovered by DRG explant cocultures from <10% of vaccinated mice compared with 100% of mock-vaccinated mice. The vaccine provided protection against disease and death after intravaginal challenge and markedly lowered the titers of the challenge virus in the vagina. Therefore, the HSV-1 gEnull strain is an excellent candidate for further vaccine development.
Figures





References
-
- Barr, E., and G. Tamms. 2007. Quadrivalent human papillomavirus vaccine. Clin. Infect. Dis. 45609-617. - PubMed
-
- Basu, S., G. Dubin, M. Basu, V. Nguyen, and H. M. Friedman. 1995. Characterization of regions of herpes simplex virus type 1 glycoprotein E involved in binding the Fc domain of monomeric IgG and in forming a complex with glycoprotein I. J. Immunol. 154260-267. - PubMed
-
- Bernstein, D. I., F. Y. Aoki, S. K. Tyring, L. R. Stanberry, C. St-Pierre, S. D. Shafran, G. Leroux-Roels, K. Van Herck, A. Bollaerts, and G. Dubin. 2005. Safety and immunogenicity of glycoprotein D-adjuvant genital herpes vaccine. Clin. Infect. Dis. 401271-1281. - PubMed
-
- Blacklaws, B. A., S. Krishna, A. C. Minson, and A. A. Nash. 1990. Immunogenicity of herpes simplex virus type 1 glycoproteins expressed in vaccinia virus recombinants. Virology 177727-736. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

