Oscillating cortical thick ascending limb cells at the juxtaglomerular apparatus
- PMID: 18562570
- PMCID: PMC2551563
- DOI: 10.1681/ASN.2007080900
Oscillating cortical thick ascending limb cells at the juxtaglomerular apparatus
Abstract
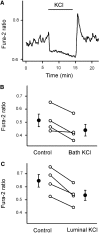
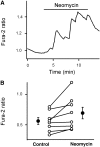
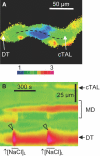
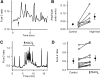
While studying the intracellular calcium dynamics in cells of the macula densa, the observation was made that tubular epithelial cells located near the macula densa and associated with the renal arterioles exhibit spontaneous Ca2+ oscillations. In this study, the cortical thick ascending limb-distal tubule, with attached glomerulus, was isolated and perfused. At a low luminal sodium chloride concentration, Ca2+ oscillations at a frequency of 63 mHz were observed in tubular cells that were within 100 microm of the macula densa plaque using four-dimensional multiphoton microscopy and wide-field fluorescence microscopy with fura-2. The Ca2+ oscillations were absent in the macula densa cells. Spontaneous oscillations in basolateral membrane potential suggested that Ca2+ oscillations occurred, at least in part, through depolarization-induced increases in Ca2+ entry. The amplitude of these Ca2+ oscillations was significantly enhanced by the activation of the Ca2+-sensing receptor. Increasing the luminal sodium chloride concentration or luminal flow resulted in a significant increase in both the amplitude of Ca2+ oscillations and the intracellular Ca2+ concentration in perimacular cortical thick ascending limb cells. In addition, luminal furosemide attenuated the [NaCl]L-dependent changes in intracellular Ca2+ concentration, but hydrochlorothiazide had no effect. These findings demonstrate that tubular epithelial cells at the perimeter of the macula densa exhibit spontaneous oscillations in intracellular Ca2+ concentration, enhanced by tubular flow and luminal sodium chloride. These oscillatory patterns may play a role in juxtaglomerular signaling.
Figures

Similar articles
-
Unraveling the relationship between macula densa cell volume and luminal solute concentration/osmolality.Kidney Int. 2006 Sep;70(5):865-71. doi: 10.1038/sj.ki.5001633. Epub 2006 Jul 5. Kidney Int. 2006. PMID: 16820788
-
Intracellular cytosolic free calcium concentration in the macula densa and in ascending limb cells at different luminal concentrations of sodium chloride and with added furosemide.Acta Physiol Scand. 1991 Jun;142(2):283-90. doi: 10.1111/j.1748-1716.1991.tb09158.x. Acta Physiol Scand. 1991. PMID: 1877377
-
Macula densa cells sense luminal NaCl concentration via furosemide sensitive Na+2Cl-K+ cotransport.Pflugers Arch. 1989 Jul;414(3):286-90. doi: 10.1007/BF00584628. Pflugers Arch. 1989. PMID: 2780213
-
Characteristics of membrane transport processes of macula densa cells.Clin Exp Pharmacol Physiol. 1997 Jul;24(7):541-7. doi: 10.1111/j.1440-1681.1997.tb01243.x. Clin Exp Pharmacol Physiol. 1997. PMID: 9248675 Review.
-
Dynamic aspects of the tubuloglomerular feedback mechanism.Dan Med Bull. 1992 Apr;39(2):134-54. Dan Med Bull. 1992. PMID: 1611920 Review.
Cited by
-
Enhancement of HLA class II-restricted CD4+ T cell recognition of human melanoma cells following treatment with bryostatin-1.Cell Immunol. 2011;271(2):392-400. doi: 10.1016/j.cellimm.2011.08.007. Epub 2011 Aug 18. Cell Immunol. 2011. PMID: 21903207 Free PMC article.
-
Electrotonic vascular signal conduction and nephron synchronization.Am J Physiol Renal Physiol. 2009 Apr;296(4):F751-61. doi: 10.1152/ajprenal.90669.2008. Epub 2008 Dec 30. Am J Physiol Renal Physiol. 2009. PMID: 19116241 Free PMC article.
-
Mechanisms of vasopressin-induced intracellular Ca2+ oscillations in rat inner medullary collecting duct.Am J Physiol Renal Physiol. 2011 Feb;300(2):F540-8. doi: 10.1152/ajprenal.00544.2009. Epub 2010 Dec 8. Am J Physiol Renal Physiol. 2011. PMID: 21147839 Free PMC article.
-
Stretch-Induced Increases in Intracellular Ca Stimulate Thick Ascending Limb O2- Production and Are Enhanced in Dahl Salt-Sensitive Rats.Hypertension. 2020 Feb;75(2):431-438. doi: 10.1161/HYPERTENSIONAHA.119.13765. Epub 2019 Dec 23. Hypertension. 2020. PMID: 31865796 Free PMC article.
-
The first decade of using multiphoton microscopy for high-power kidney imaging.Am J Physiol Renal Physiol. 2012 Jan 15;302(2):F227-33. doi: 10.1152/ajprenal.00561.2011. Epub 2011 Oct 26. Am J Physiol Renal Physiol. 2012. PMID: 22031850 Free PMC article. Review.
References
-
- Golgi C: Notes on the histology of human and mammalian kidney and on the histogenesis of the nephron. Atti della Reale Accademia dei Lincei 5: 334–342, 1889
-
- Schnermann J, Briggs JP: Function of the juxtaglomerular apparatus: Control of glomerular hemodynamics and renin secretion. In: The Kidney Physiology & Pathophysiology, 3rd Ed., edited by Seldin DW, Giebisch G, Philadelphia, Lippincott Williams & Wilkins, 2000, pp 945–980
-
- Barajas L, Salido EC, Liu L, Powers KV: The juxtaglomerular apparatus: A morphologic perspective. In: Hypertension: Pathophysiology, Diagnosis, and Management, 2nd Ed., edited by Laragh JH, Brenner BM, New York, Raven Press, Ltd., 1995, pp 1335–1348
-
- Barajas L: Anatomy of the juxtaglomerular apparatus. Am J Physiol 237: F333–F343, 1979 - PubMed
-
- Riccardi D, Hall AE, Chattopadhyay N, Xu JZ, Brown EM, Hebert SC: Localization of the extracellular Ca2+/polyvalent cation-sensing protein in rat kidney. Am J Physiol 274: F611–F622, 1998 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

